Introduction
Most conversations around thyroid dysfunction focus on hormone levels, medication, and symptom management. Yet emerging systems biology research shows that thyroid physiology does not operate in isolation. Hormone production in the thyroid gland is only one part of a much larger regulatory network that includes immune signalling, liver metabolism, and increasingly, the gut microbiome.
The concept of the thyroid-gut axis reflects a deeper biological reality: intestinal microbes participate directly in hormone activation, immune tolerance, nutrient absorption, and inflammatory regulation. When microbial balance is disrupted, the effects extend far beyond digestion. Hormone conversion efficiency declines, immune surveillance shifts, and inflammatory signals begin interfering with endocrine communication.
This is why individuals may present with persistent thyroid symptoms despite “normal thyroid levels” on laboratory testing. The gland may be producing hormones, but downstream activation and cellular utilisation may be compromised by gut dysfunction.
Understanding the microbiome and thyroid connection helps explain rising thyroid disorders, especially autoimmune thyroiditis and metabolic thyroid resistance. It also clarifies why comprehensive thyroid treatment must extend beyond the gland itself.
The Thyroid Is Not an Isolated Organ
Hormone Production vs Hormone Activation
The thyroid gland primarily produces thyroxine (T4), a storage hormone. Only a small fraction of circulating hormones exists as triiodothyronine (T3), the metabolically active form that regulates cellular energy production, temperature regulation, and metabolic signalling.
Activation from T4 to T3 is not controlled solely by the thyroid gland. It depends on deiodinase enzymes distributed throughout the body and particularly in the liver, peripheral tissues, and importantly, the gastrointestinal system.
A significant portion of thyroid hormone metabolism occurs through enterohepatic circulation. Hormones are conjugated in the liver, secreted into bile, and then processed again by intestinal bacteria. Specific microbial enzymes help deconjugate hormone metabolites, allowing them to be reabsorbed and reused.
When microbial diversity is reduced, this recycling pathway becomes inefficient. Hormone clearance may increase, active hormone availability declines, and metabolic signalling weakens even when hormone production appears adequate.
This explains why gut dysbiosis and hypothyroidism frequently coexist, not merely as parallel conditions but as mechanistically linked processes.
Immune Regulation Begins in the Gut
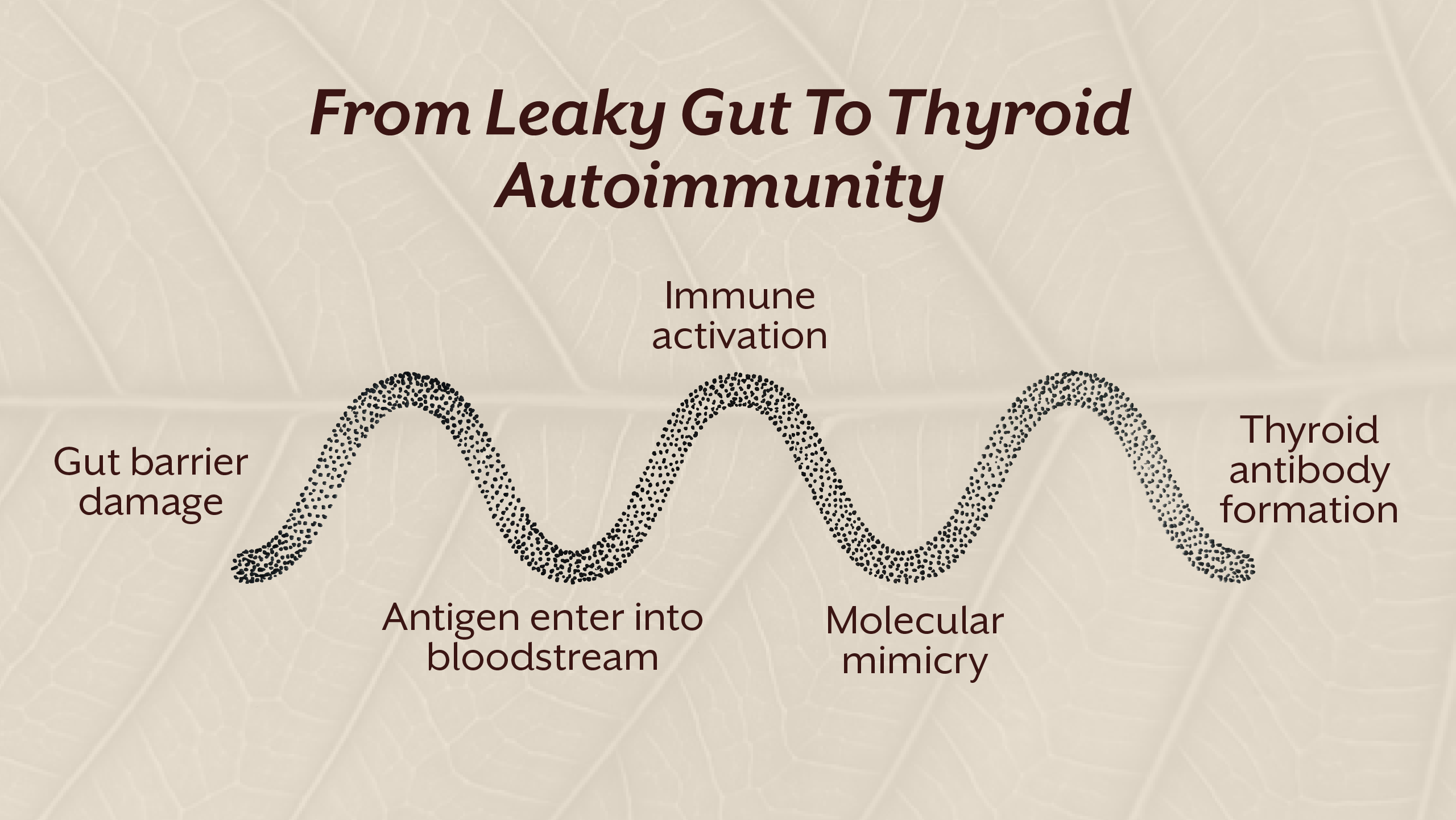
Nearly seventy percent of immune activity originates within the gastrointestinal system. The intestinal barrier acts as both a physical and immunological filter, determining which molecules enter systemic circulation.
When microbial balance is stable, immune tolerance is maintained. Regulatory immune cells prevent unnecessary inflammatory responses, protecting self-tissues including endocrine glands.
When dysbiosis develops, inflammatory signalling increases. Gut permeability rises. Antigen exposure expands. Immune regulation becomes unstable.
This environment increases the likelihood of thyroid antibodies forming, a defining feature of autoimmune thyroid disorders such as Hashimoto’s thyroiditis and other forms of thyroiditis.
Microbiome and Thyroid Hormone Conversion
Bacterial Enzymes and Hormone Reactivation
Certain intestinal bacteria produce enzymes such as beta-glucuronidase that influence hormone reactivation. These enzymes regulate how efficiently conjugated thyroid hormones are reabsorbed rather than eliminated.
When microbial diversity declines, enzyme activity becomes unpredictable. Hormones may be excessively eliminated or improperly reactivated. The result is functional hormone deficiency at the tissue level.
This is one of the lesser discussed thyroid causes meaning altered microbial enzyme activity affecting endocrine signalling.
Gut Inflammation and Reverse T3 Formation
Inflammatory stress alters hormone conversion pathways. Instead of converting T4 into active T3, the body may increase production of reverse T3, an inactive molecule that blocks receptor activity.
Gut inflammation is a major contributor to this shift. Inflammatory cytokines suppress deiodinase activity while activating stress signalling pathways that favour energy conservation.
This physiological adaptation may be protective during acute illness. However, chronic gut inflammation can sustain high reverse T3 levels, slowing metabolism despite adequate hormone production.
Nutrient Absorption and Thyroid Hormone Function
Thyroid hormone synthesis and activation depend heavily on micronutrients such as selenium, zinc, iodine, iron, and tyrosine. These nutrients are absorbed primarily in the gastrointestinal tract.
Dysbiosis reduces absorption efficiency. Inflammatory changes impair transport mechanisms. Enzymatic digestion becomes inconsistent.
Even with an optimal diet, individuals with gut imbalance may develop functional nutrient deficiency, impairing thyroid hormone function and contributing to persistent thyroid disease symptoms.

Microbiome and Thyroid Hormone Conversion
Bacterial Enzymes and Hormone Reactivation
Certain intestinal bacteria produce enzymes such as beta-glucuronidase that influence hormone reactivation. These enzymes regulate how efficiently conjugated thyroid hormones are reabsorbed rather than eliminated.
When microbial diversity declines, enzyme activity becomes unpredictable. Hormones may be excessively eliminated or improperly reactivated. The result is functional hormone deficiency at the tissue level.
This is one of the lesser discussed thyroid causes meaning altered microbial enzyme activity affecting endocrine signalling.
Gut Inflammation and Reverse T3 Formation
Inflammatory stress alters hormone conversion pathways. Instead of converting T4 into active T3, the body may increase production of reverse T3, an inactive molecule that blocks receptor activity.
Gut inflammation is a major contributor to this shift. Inflammatory cytokines suppress deiodinase activity while activating stress signalling pathways that favour energy conservation.
This physiological adaptation may be protective during acute illness. However, chronic gut inflammation can sustain high reverse T3 levels, slowing metabolism despite adequate hormone production.
Nutrient Absorption and Thyroid Hormone Function
Thyroid hormone synthesis and activation depend heavily on micronutrients such as selenium, zinc, iodine, iron, and tyrosine. These nutrients are absorbed primarily in the gastrointestinal tract.
Dysbiosis reduces absorption efficiency. Inflammatory changes impair transport mechanisms. Enzymatic digestion becomes inconsistent.
Even with an optimal diet, individuals with gut imbalance may develop functional nutrient deficiency, impairing thyroid hormone function and contributing to persistent thyroid disease symptoms.
Why Conventional Thyroid Treatment Often Falls Short
Standard thyroid treatment typically focuses on hormone replacement or suppression depending on clinical presentation. While this approach can stabilize circulating hormone levels, it does not address upstream regulatory disturbances.
If gut inflammation persists, conversion pathways remain impaired. If microbial imbalance continues, immune activation remains elevated. If nutrient absorption is compromised, hormone utilisation remains inefficient.
Patients may therefore achieve laboratory normalisation without full physiological restoration.
This explains why many individuals with thyroid symptoms continue experiencing fatigue, metabolic slowing, and immune activation despite treatment.
A Systems Approach to Thyroid Health
A clinically effective thyroid strategy must address the entire regulatory network.
Interventions targeting microbial diversity, intestinal barrier repair, inflammatory modulation, and micronutrient repletion influence hormone conversion more profoundly than gland-focused interventions alone.
This systems-biology perspective is central to the clinical model used at iThrive Alive, where thyroid physiology is evaluated as part of an integrated metabolic and immune framework.
Rather than isolating the gland, assessment includes gut health, inflammatory load, nutrient status, and stress physiology. This enables identification of root drivers behind hormone dysregulation.
Individuals seeking deeper evaluation can book a root cause analysis or book a consult to explore underlying regulatory imbalances rather than focusing solely on hormone levels.

Key Takeaway
The thyroid-gut axis represents one of the most clinically significant yet under-recognised regulatory pathways in endocrine physiology. Hormone production within the thyroid gland is only the beginning of metabolic signalling. Activation, recycling, immune tolerance, and cellular response depend heavily on microbial balance, intestinal integrity, and inflammatory regulation. When gut dysbiosis develops, hormone conversion shifts, reverse T3 increases, immune reactivity intensifies, and nutrient availability declines. These processes can sustain thyroid dysfunction even when laboratory values appear stable. Understanding thyroid physiology through the lens of systems biology reveals why comprehensive treatment must extend beyond hormone replacement toward restoration of microbial balance, immune stability, and metabolic resilience.


.svg)
.svg)























