Introduction
Many men silently struggle with erectile dysfunction or low sexual drive but rarely talk about it. When it does come up, the discussion often turns immediately toward medications or hormonal problems. However, there is an important biological layer that is frequently ignored. The health of the tiny energy factories inside our cells.
Erectile dysfunction, also known as impotence, is not only about hormones or aging. It is deeply connected to cellular metabolism, blood vessel function, nerve signaling, and energy production inside the body. All of these systems rely heavily on mitochondrial health.
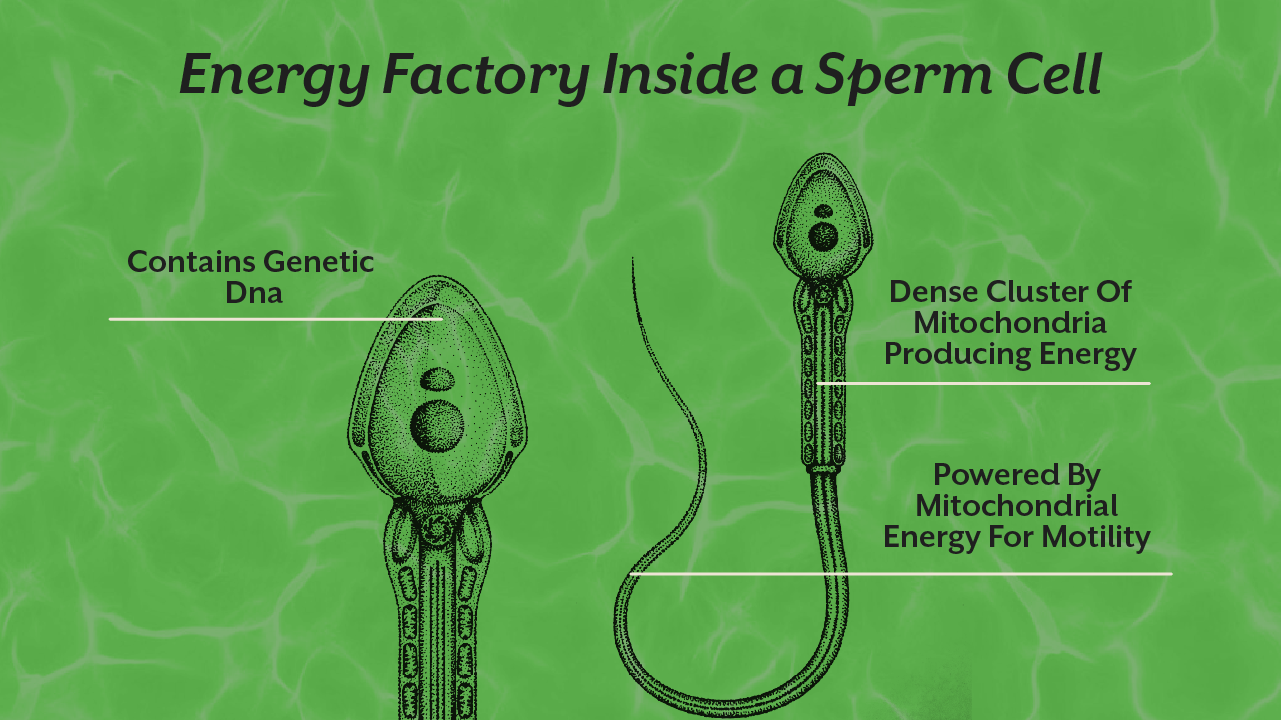
Mitochondria are responsible for producing ATP, the energy currency that powers almost every process in the body. From muscle contraction to hormone production and blood vessel relaxation, mitochondria quietly control the physiological processes that make a healthy erection possible.
When mitochondrial function declines due to poor nutrition, chronic inflammation, metabolic stress, or insulin resistance, the body begins to lose efficiency in multiple systems. Blood vessels become less responsive, nerve signaling slows down, and hormonal balance may shift. Over time this can appear as erectile dysfunction.
For many men, erectile dysfunction is therefore not simply a sexual health issue. It may be an early metabolic signal that something deeper in the body needs attention.
Understanding this connection can completely change the way we approach erectile dysfunction and the strategies used to restore health.
Understanding Erectile Dysfunction Beyond Hormones
Erectile dysfunction occurs when a man consistently finds it difficult to achieve or maintain an erection sufficient for sexual activity. While testosterone is often blamed for this issue, the physiology of erection is actually much more complex.
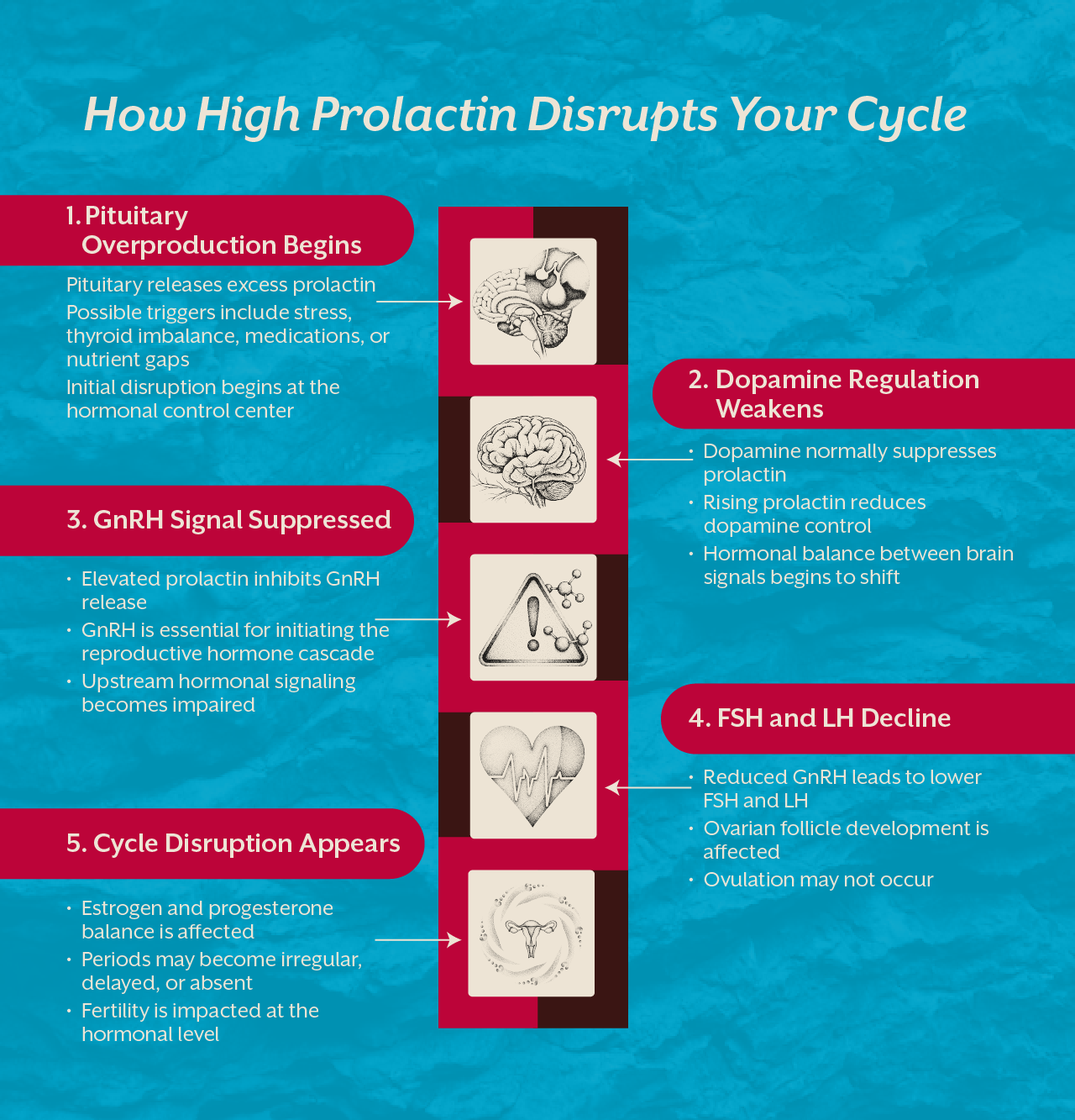
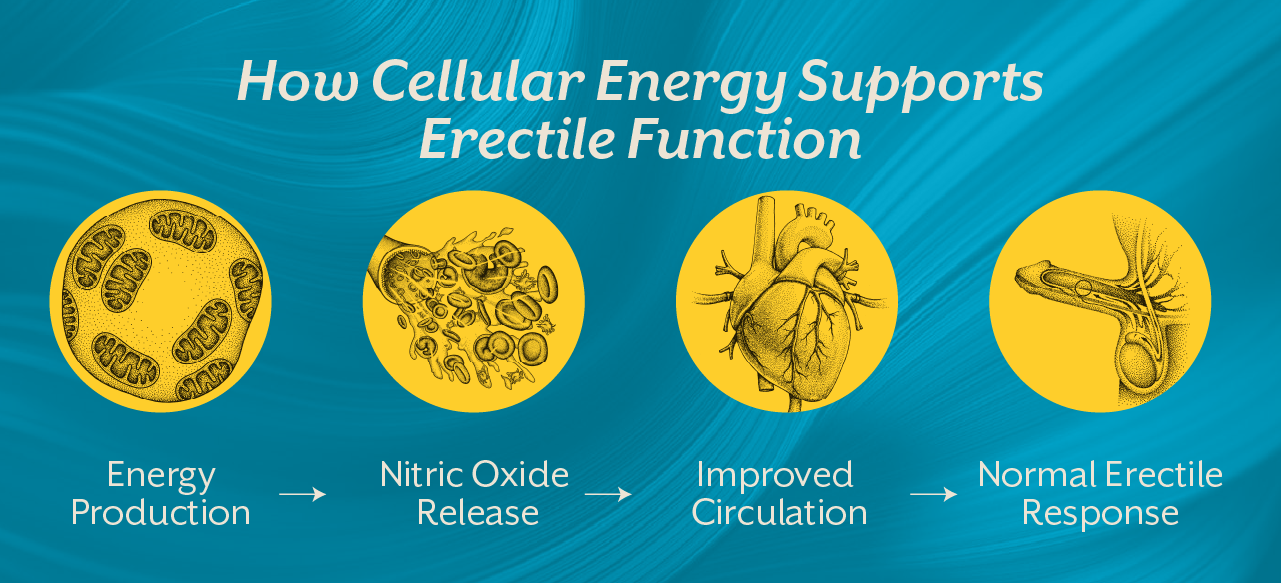
An erection depends on a coordinated interaction between blood vessels, nerve signals, hormones, and cellular metabolism. When sexual stimulation occurs, nerve signals trigger the release of nitric oxide in penile blood vessels. Nitric oxide relaxes smooth muscles and allows blood to flow into the erectile tissues.
For this process to work smoothly, blood vessels must respond properly, nerves must transmit signals efficiently, and cells must produce sufficient energy to sustain these responses.
This is where mitochondrial function becomes critically important.
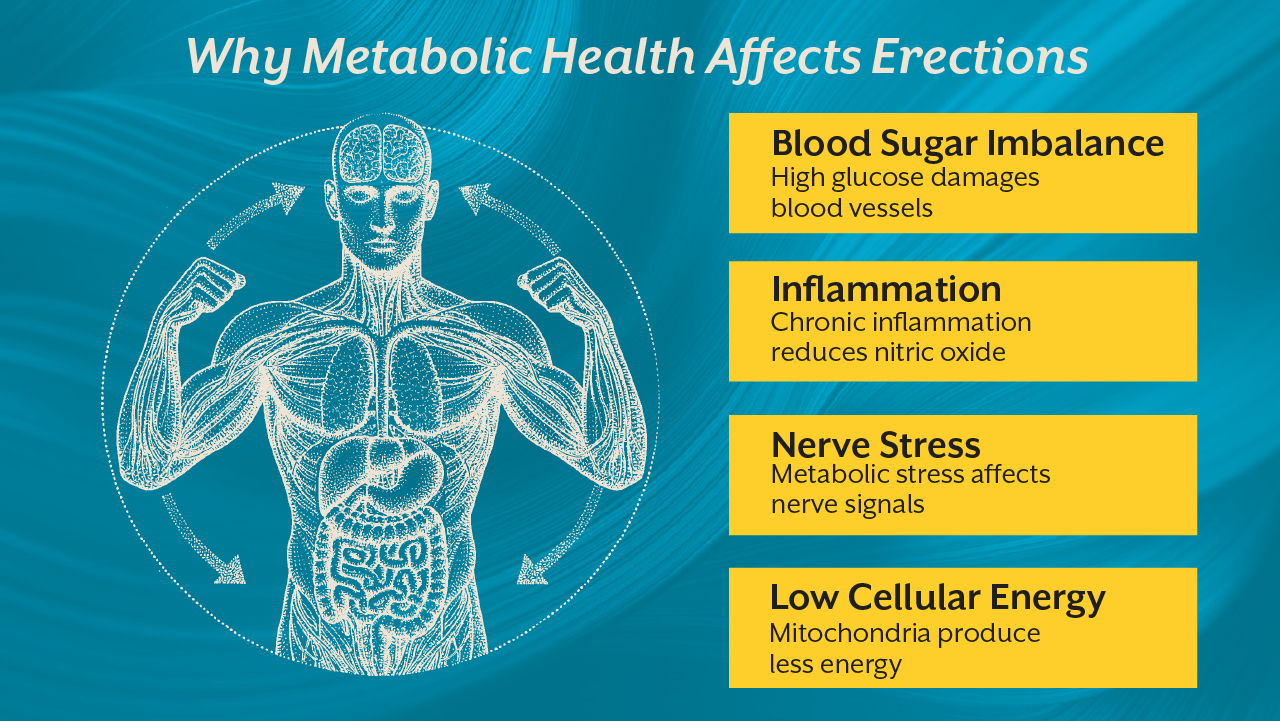
Mitochondria regulate cellular energy production and help maintain the integrity of blood vessel lining known as the endothelium. When mitochondrial function is compromised, nitric oxide signaling weakens and blood vessels lose their ability to dilate effectively. The result is impaired circulation, which directly affects erectile response.
This is why erectile dysfunction is increasingly considered an early marker of metabolic and vascular health issues.
How Mitochondrial Dysfunction Affects Erectile Health
Mitochondria play a surprisingly large role in reproductive physiology. They are involved not only in energy production but also in regulating oxidative stress, hormone synthesis, and vascular health.
When mitochondrial efficiency declines, several biological disruptions begin to appear.
First, reduced ATP production limits the ability of blood vessels to relax and expand. This directly affects the blood flow required to maintain an erection.
Second, mitochondrial dysfunction increases oxidative stress. Excess oxidative stress damages endothelial cells, which are responsible for producing nitric oxide. Without adequate nitric oxide signaling, blood vessels cannot dilate effectively.
Third, mitochondria play an important role in steroid hormone metabolism. Testosterone synthesis involves several mitochondrial steps. If mitochondrial activity is impaired, hormonal balance may gradually decline.
Finally, mitochondrial dysfunction contributes to chronic inflammation and metabolic disorders. Conditions such as insulin resistance and metabolic syndrome are strongly associated with erectile dysfunction.
When viewed through this lens, erectile dysfunction becomes less about isolated reproductive organs and more about systemic metabolic health.
The Metabolic Link Between Diabetes And Erectile Dysfunction
One of the strongest examples of metabolic influence on erectile function is seen in diabetes. Men with diabetes are significantly more likely to experience erectile dysfunction compared to the general population.
Chronic high blood sugar creates oxidative stress that damages both blood vessels and nerve fibers. This leads to endothelial dysfunction and impaired nerve signaling. Both are essential components of erectile physiology.
In addition, insulin resistance reduces mitochondrial efficiency. When cells struggle to process glucose properly, energy production becomes inefficient and inflammatory signaling increases. Over time this metabolic environment disrupts vascular responsiveness.
Many men first notice erectile changes years before diabetes is formally diagnosed. This makes erectile dysfunction an early metabolic warning sign.
If you want to understand how metabolic health influences chronic disease risk, the iThrive Alive blog titled Insulin Resistance: The Silent Phase Before Type 2 Diabetes explains how metabolic dysfunction develops long before clinical diagnosis.
What You Can Do: Nutrition And Lifestyle That Support Mitochondrial Health
The encouraging news is that mitochondrial function is highly responsive to lifestyle interventions. Unlike many structural diseases, mitochondrial efficiency can often improve when the right metabolic environment is created.
At iThrive Alive, the focus is on addressing the root drivers of dysfunction rather than only suppressing symptoms.
One of the first areas addressed is nutrition. A diet that stabilizes blood sugar and reduces inflammatory stress allows mitochondria to function more efficiently. Whole foods rich in micronutrients support enzymatic pathways involved in cellular energy production.
Smart supplementation can further assist mitochondrial function. Nutrients such as magnesium, zinc, CoQ10, and carnitine support mitochondrial respiration and antioxidant defense systems.
Lifestyle interventions also play a significant role. Regular movement improves mitochondrial density in muscle tissue and enhances insulin sensitivity. Sleep quality influences hormone regulation and mitochondrial repair processes.
Stress management is equally important because chronic stress hormones impair mitochondrial metabolism and contribute to systemic inflammation.
These changes may appear simple, but their cumulative impact on metabolic health can be profound.

Why A Root Cause Approach Matters
Conventional management of erectile dysfunction often focuses on medications that temporarily improve blood flow. While these medications can be helpful for symptom relief, they do not address the metabolic environment that created the problem.
A root cause approach looks deeper. It examines factors such as insulin resistance, inflammation, mitochondrial health, hormonal balance, and nutrient status.
At iThrive Alive, this is where personalized metabolic assessment becomes valuable. Through detailed evaluation and targeted interventions, the goal is to identify the biological drivers behind symptoms and support long term physiological recovery.
For individuals experiencing persistent erectile dysfunction or unexplained low drive, it may be helpful to book a root cause analysis to understand the metabolic factors involved.
When Erectile Dysfunction Is A Signal From Your Body
Many men hesitate to discuss erectile dysfunction because it feels personal and uncomfortable. Yet from a biological perspective, the body is often sending an early signal that metabolic health requires attention.
Blood vessels, hormones, and nerves are deeply connected to overall metabolic function. When these systems begin to show strain, erectile changes may appear long before other symptoms become obvious.
Rather than viewing erectile dysfunction as an isolated problem, it can be helpful to see it as an opportunity to improve long term health.
If you are experiencing these symptoms and want guidance, you can also book a consult with the nutritionist to explore personalized strategies for improving metabolic and reproductive health.
Key Takeaway
Erectile dysfunction is rarely just about sexual health. It reflects a complex interaction between vascular function, nerve signaling, hormonal balance, and cellular energy metabolism. Mitochondrial dysfunction sits at the center of this network because every step of erectile physiology depends on adequate cellular energy production. When mitochondria struggle due to poor nutrition, metabolic stress, or inflammation, the systems responsible for erectile function begin to lose efficiency. By focusing on metabolic health through nutrition, lifestyle changes, and targeted interventions, it is possible to support mitochondrial recovery and improve overall physiological resilience. Addressing the root cause not only supports reproductive health but also strengthens long term metabolic wellbeing.


.svg)
.svg)