Introduction
Prediabetes is often presented as a mild elevation in blood sugar, a temporary phase between “normal” and diabetes. This framing is misleading and, in many cases, dangerously incomplete. Prediabetes is not a glucose problem waiting to become severe. It is a metabolic warning signal indicating that the body’s energy regulation systems are already under strain.
Most individuals diagnosed with prediabetes are told to “watch sugar,” “eat less carbs,” or “exercise more.” While these suggestions are not incorrect, they fail to address the underlying physiology driving the condition. Blood sugar rises only when the body can no longer compensate for deeper metabolic dysfunction, particularly insulin resistance.
Understanding what prediabetes truly represents requires shifting focus away from sugar alone and toward metabolic health as a whole. This blog will explain the real meaning of prediabetes, why it develops, how insulin resistance sits at its core, and why early metabolic screening matters far more than waiting for diabetes to appear.
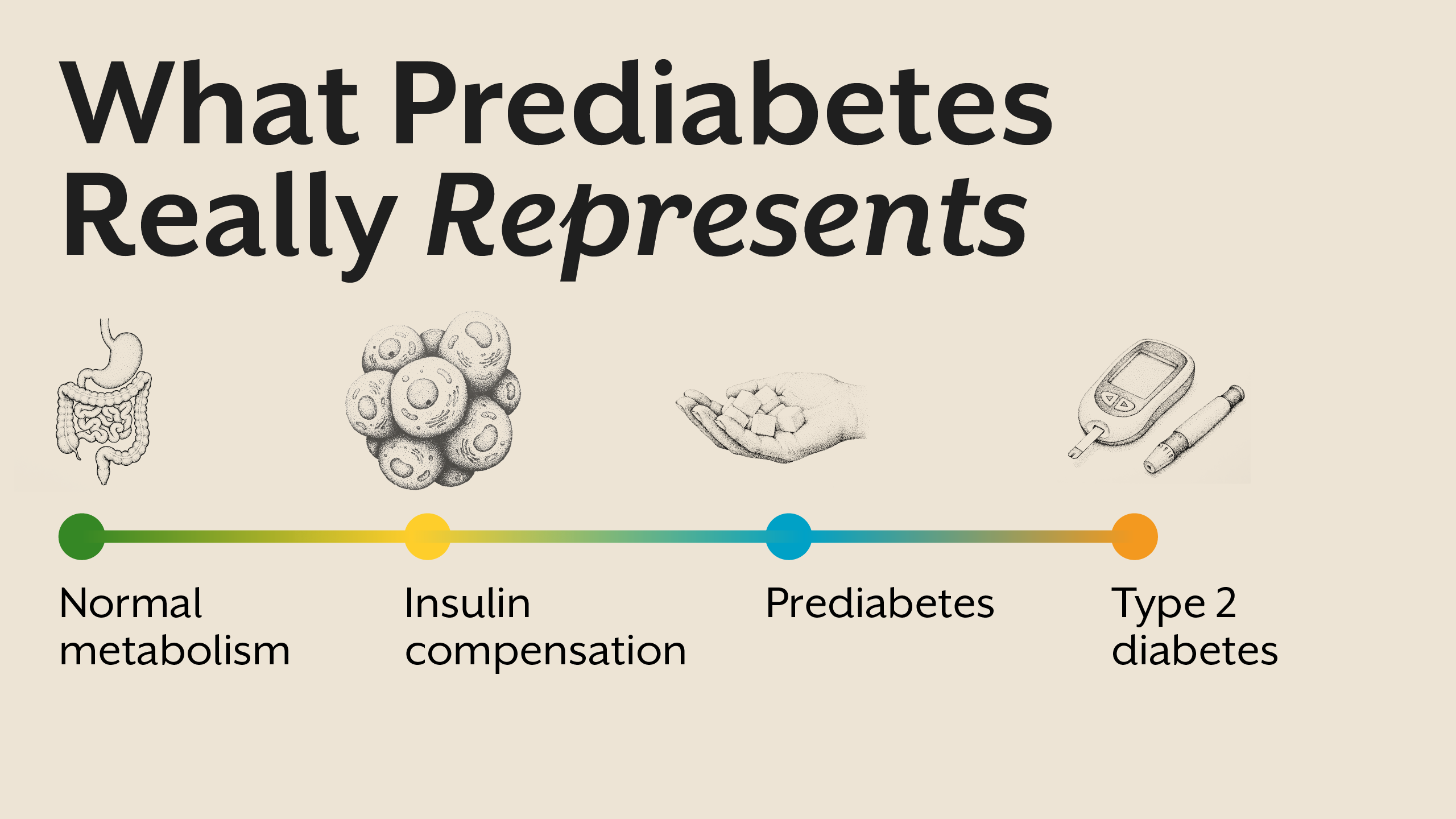
What Is Prediabetes? Understanding the Real Meaning
Prediabetes meaning is often simplified to “slightly high blood sugar.” Clinically, it is defined by fasting glucose, post-meal glucose, or HbA1c values that fall above normal but below diabetic thresholds. However, these numbers reflect outcomes, not causes.
Prediabetes represents a state where the body is struggling to maintain glucose balance despite increasing hormonal effort. Insulin levels are often elevated, signaling that cells are becoming less responsive. Blood sugar rises not because sugar intake suddenly increases, but because insulin’s effectiveness is declining.
In this phase, glucose may still appear controlled at rest, but metabolic flexibility is impaired. The body has lost its ability to adapt efficiently to meals, fasting, stress, and physical activity. Prediabetes is therefore not a borderline condition; it is an early disease state.
Why Prediabetes Is Not Caused by Sugar Alone
Sugar does not create prediabetes in isolation. If it did, every individual consuming carbohydrates would develop the condition. The real issue lies in how the body processes and responds to glucose.
Insulin resistance is the central driver. When cells fail to respond efficiently to insulin, glucose remains in circulation longer, prompting the pancreas to release more insulin. Over time, this compensatory mechanism becomes insufficient.
Additional contributors include chronic stress, sleep deprivation, circadian disruption, inflammation, micronutrient deficiencies, and frequent eating without metabolic rest. These factors collectively impair insulin signaling and mitochondrial efficiency.
Prediabetes Symptoms: Often Subtle, Rarely Recognized
Prediabetes symptoms are not dramatic. Most individuals do not feel “unwell” enough to seek medical attention. Instead, they experience gradual changes that are normalized or ignored.
Common symptoms include persistent fatigue, post-meal drowsiness, difficulty losing weight, increased hunger, sugar cravings, and abdominal fat gain. Cognitive symptoms such as brain fog and reduced focus are also common.
Because blood sugar levels are only mildly elevated, these symptoms are rarely linked to metabolic dysfunction. Yet they reflect a body working harder to maintain balance.
Insulin Resistance: The Core of Prediabetes
Insulin resistance sits at the heart of prediabetes. It develops when cells in muscle, liver, and adipose tissue stop responding effectively to insulin’s signal. As resistance increases, insulin levels rise to compensate.
This process is not uniform. Muscle tissue may resist insulin first, impairing glucose uptake. The liver may continue producing glucose despite insulin presence. Adipose tissue may remain insulin sensitive, promoting fat storage.
These tissue-specific responses explain why individuals with prediabetes often gain visceral fat while still being told their sugar is “borderline.” Insulin resistance reflects systemic metabolic dysfunction, not a single lab value.

Prediabetes Causes: A Systems-Level Breakdown
Prediabetes causes are cumulative, not isolated. Frequent eating keeps insulin elevated throughout the day. Chronic stress elevates cortisol, worsening glucose production and insulin resistance. Poor sleep impairs insulin signaling and mitochondrial function.
Circadian disruption from late nights and irregular meals further destabilizes metabolic rhythms. Nutrient deficiencies, particularly magnesium and B-complex vitamins, impair glucose metabolism at the cellular level.
Prediabetes reflects a system under constant demand without sufficient recovery. Addressing it requires restoring metabolic rhythm, not simply lowering sugar intake.
Prediabetes Treatment: Shifting the Focus
Prediabetes treatment is often framed as glucose reduction. However, targeting sugar alone ignores the hormonal dysfunction driving the condition.
Effective intervention focuses on improving insulin sensitivity, reducing insulin demand, restoring circadian alignment, and supporting mitochondrial health. Meal timing, sleep quality, stress regulation, and nutrient sufficiency play central roles.
Strategic fasting, when individualized, allows insulin levels to normalize and receptors to resensitize. Physical activity improves glucose uptake independent of insulin. These approaches address root causes rather than symptoms.

Why Prediabetes Is a Window, Not a Waiting Room
Prediabetes is often treated as a warning label with no urgency. In reality, it is a narrow window where metabolic flexibility can still be restored.
Once insulin resistance progresses and pancreatic capacity declines, reversal becomes significantly harder. Early identification allows targeted intervention before irreversible damage occurs.
This is why Alive emphasizes early metabolic screening and Root Cause Analysis rather than waiting for diagnostic thresholds. If you are uncertain whether prediabetes reflects deeper metabolic dysfunction, you may consider booking a free consult to understand whether further evaluation is needed.
Prediabetes and Metabolic Health: The Bigger Picture
Prediabetes is not an isolated diagnosis. It reflects declining metabolic health across systems. Improving metabolic health improves glucose regulation as a downstream effect.
This broader view aligns with Essentials philosophy, where foundational metabolic support precedes symptom management. When insulin sensitivity improves, glucose stability often follows naturally.
Key Takeaway
Prediabetes is not a mild sugar abnormality but an early warning of metabolic dysfunction driven primarily by insulin resistance. Blood glucose rises only after the body has exhausted years of hormonal compensation, making prediabetes a critical window for intervention rather than a passive waiting phase. Addressing metabolic health through insulin sensitivity, lifestyle rhythm, and root cause evaluation allows meaningful reversal before long-term disease develops.


.svg)
.svg)















.webp)