



.svg)




.svg)
When most people hear the words rheumatoid arthritis, they imagine swollen joints, stiff fingers, and persistent pain. The disease is often described as a joint condition where the immune system mistakenly attacks the lining of joints. This explanation is technically correct but biologically incomplete.
Many individuals diagnosed with rheumatoid arthritis report something else long before joint swelling appears. They experience fatigue, digestive discomfort, brain fog, poor recovery after exertion, or unexplained inflammation across the body. These symptoms rarely seem connected to arthritis at first. Yet in many cases they are the earliest signs that something deeper is happening.
Autoimmune arthritis does not begin inside the joints alone. It reflects systemic changes within the immune system, metabolism, and cellular energy networks. One emerging driver receiving growing scientific attention is mitochondrial dysfunction. Mitochondria are the cellular structures responsible for generating energy inside every cell of the body. When these energy systems begin to fail, immune regulation changes and inflammation increases.
This shift can quietly create the biological environment that later manifests as rheumatoid arthritis symptoms.
Understanding these hidden mechanisms opens a different perspective on RA treatment. Instead of focusing solely on suppressing symptoms, it becomes possible to examine the deeper biological conditions that allowed the disease to emerge.
Rheumatoid arthritis is classified as an autoimmune arthritis disorder in which the immune system mistakenly targets healthy tissue. The most visible target becomes the synovial membrane that surrounds joints. Inflammation within this tissue produces swelling, stiffness, and joint pain.
However the immune system does not operate in isolation from the rest of the body. Immune behavior is heavily influenced by metabolic signals, cellular energy production, and nutrient availability.
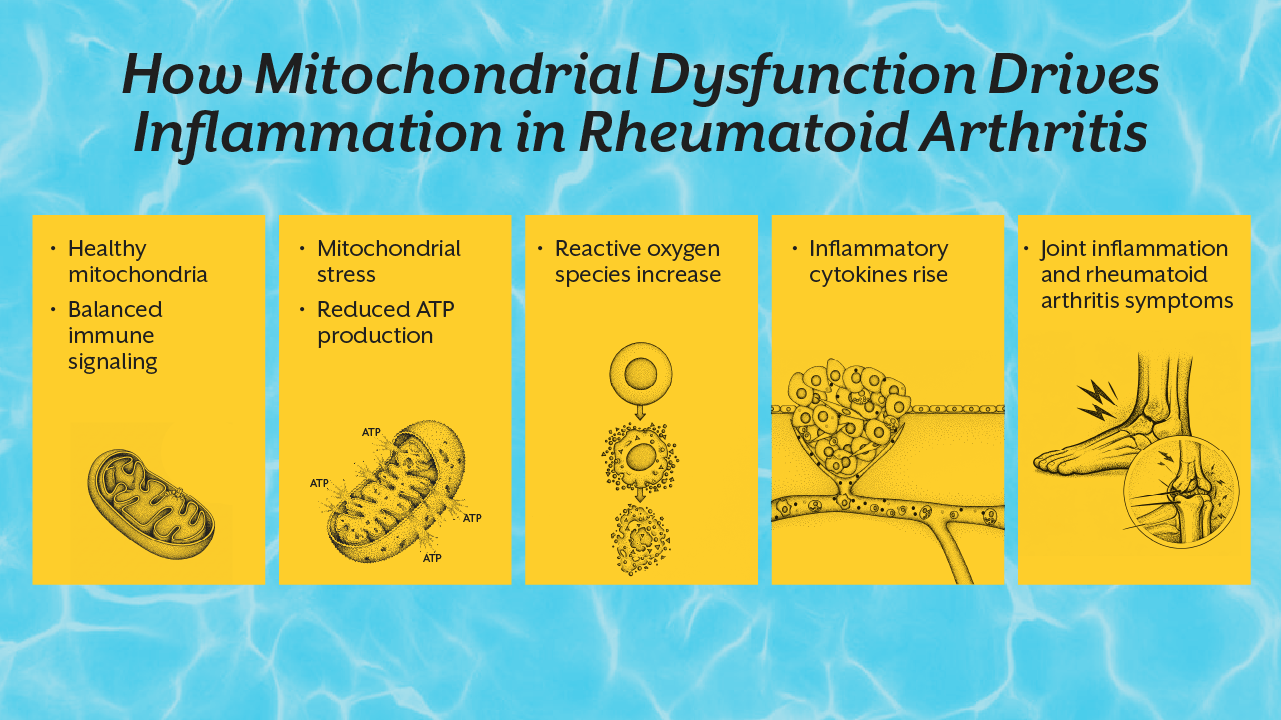
In many individuals with rheumatoid arthritis symptoms, researchers have identified disturbances in mitochondrial metabolism within immune cells. When mitochondrial energy production declines, immune cells shift toward a more inflammatory state.
This shift activates inflammatory pathways that release cytokines such as TNF alpha and interleukin six. These molecules amplify inflammation in rheumatoid arthritis and contribute to joint damage over time.
The joints therefore become the visible site of inflammation. But the origin of the immune disturbance often lies deeper within the metabolic systems of the body.
To understand why mitochondrial health matters in rheumatoid arthritis, it helps to examine how immune cells generate energy.
Immune cells rely on mitochondria to produce ATP which powers cellular activity. When mitochondria function normally, immune responses remain balanced and regulated.
However mitochondrial dysfunction can shift immune metabolism toward a state known as inflammatory metabolic reprogramming. In this state immune cells rely more heavily on glycolysis rather than efficient mitochondrial respiration. This metabolic change promotes excessive inflammatory signaling.
Several studies have identified mitochondrial abnormalities within synovial cells of individuals with rheumatoid arthritis. These cells show signs of oxidative stress, impaired mitochondrial respiration, and reduced energy production. The consequences extend beyond the joints.
Low cellular energy also contributes to fatigue, reduced physical resilience, and slow tissue repair. Many individuals with autoimmune arthritis report persistent exhaustion long before joint damage becomes severe.
This suggests that mitochondrial dysfunction may act as an early driver that shapes the inflammatory environment in rheumatoid arthritis.
Another hidden driver of inflammation in rheumatoid arthritis involves metabolic signals that influence immune activity.
Many individuals with autoimmune arthritis demonstrate metabolic abnormalities including impaired glucose regulation, oxidative stress, and altered lipid metabolism. These metabolic disturbances produce inflammatory signals that can worsen autoimmune reactions.
Inflammation in rheumatoid arthritis is therefore not only the result of immune malfunction. It is also shaped by the metabolic environment in which immune cells operate.
Mitochondrial stress plays a central role in this process. When mitochondria struggle to generate energy efficiently they release reactive oxygen species. These molecules activate inflammatory pathways that amplify immune activity. Over time this environment increases tissue vulnerability and accelerates joint inflammation.
This is one reason arthritis pain management strategies that focus only on pain suppression rarely address the full biological picture.

One of the most confusing aspects of rheumatoid arthritis symptoms is the profound fatigue experienced by many patients.
Fatigue in autoimmune arthritis is frequently dismissed as a side effect of chronic pain. However metabolic research suggests a different explanation.
When mitochondrial energy production declines, tissues throughout the body receive less cellular energy. Muscles, brain cells, and immune cells all depend on mitochondrial ATP production. Reduced ATP availability leads to widespread exhaustion.
This fatigue can appear years before significant joint damage develops. It may be accompanied by brain fog, poor exercise tolerance, or prolonged recovery after physical activity.
These early signs suggest that rheumatoid arthritis may be part of a broader systemic energy imbalance rather than a disease that begins solely in the joints.
Understanding these hidden drivers opens the door to a broader view of RA treatment.
Traditional approaches often focus primarily on suppressing inflammation through medications. While these treatments can reduce symptoms, they may not address the metabolic conditions that influence immune behavior.
A systems approach examines the body as an interconnected network where immune function, mitochondrial energy production, and metabolic health influence one another. At iThrive Alive this perspective guides the evaluation process used during a root cause analysis. Rather than focusing only on the joints, the analysis explores deeper biological systems including metabolic function, nutrient status, inflammatory markers, and cellular energy metabolism.
This type of evaluation can help identify factors that may contribute to ongoing inflammation in rheumatoid arthritis. For individuals navigating autoimmune arthritis, understanding these underlying influences can provide valuable insight into their condition.
Mitochondrial function is highly sensitive to lifestyle patterns.
Sleep quality, dietary composition, physical activity, and environmental stressors all influence mitochondrial health. When these factors are optimized, mitochondrial energy production can improve and inflammatory signaling may decline. Many individuals with rheumatoid arthritis benefit from approaches that support cellular energy metabolism. These strategies may include anti inflammatory dietary patterns, nutrient repletion, and targeted supplementation that supports mitochondrial pathways.
Some supplements for rheumatoid arthritis are explored for their ability to influence oxidative stress and mitochondrial efficiency. While supplements alone cannot treat autoimmune arthritis, they may play a supportive role when combined with broader lifestyle strategies.
Within the iThrive Alive framework these interventions are implemented through the structured 3 months Alive program, which focuses on metabolic restoration through lifestyle interventions, smart supplementation, and dietary protocols. The goal is not simply arthritis pain management. The deeper objective is restoring the biological environment in which the immune system operates.

Rheumatoid arthritis is often described as a disease of the joints, but the biology behind the condition is far more complex. The immune system does not operate independently from metabolism or cellular energy systems. Emerging research suggests that mitochondrial dysfunction may play a significant role in shaping the inflammatory environment associated with autoimmune arthritis. When cellular energy systems begin to struggle, immune regulation changes and inflammatory signaling increases. Over time this environment can contribute to the development and progression of rheumatoid arthritis symptoms. By exploring the deeper metabolic drivers behind inflammation, it becomes possible to view RA treatment through a broader systems perspective that examines immune health, energy metabolism, and lifestyle factors together.



.svg)
Multiple sclerosis is often described as a neurological disease where the immune system attacks the brain and spinal cord. For decades the focus of research and treatment has been centered almost entirely on the nervous system. Yet emerging evidence suggests that the earliest drivers of this condition may begin far from the brain.
Many researchers are now exploring a deeper question. What if the biological origins of multiple sclerosis start in the gut.
The human digestive system is not only responsible for breaking down food. It also houses trillions of microorganisms, regulates immune responses, and communicates continuously with the brain through what scientists call the gut brain axis.
When this communication system is disrupted, inflammation, immune dysfunction, and metabolic stress can develop across the body. In individuals with the multiple sclerosis disease, these systemic disturbances may create conditions that allow the immune system to mistakenly attack the protective myelin sheath surrounding nerve fibers.
Understanding the causes of MS therefore requires looking beyond the nervous system and exploring the metabolic and microbial environment of the entire body.
This broader perspective is central to the clinical model used at iThrive Alive, where chronic illness is approached through a systems biology framework rather than an organ isolated view.
Multiple sclerosis is classified as an autoimmune condition of the central nervous system. In this MS disorder the immune system mistakenly attacks the myelin sheath that surrounds nerve fibers. Myelin acts as an insulating layer that allows electrical signals to travel rapidly between the brain and the rest of the body.
When myelin is damaged, communication between nerve cells slows down or becomes disrupted. Over time this can lead to symptoms such as muscle weakness, coordination problems, fatigue, and cognitive changes.
While these symptoms appear neurological, the biological processes driving them often involve immune dysfunction and chronic inflammation.
Many individuals diagnosed with the MS medical condition also show systemic imbalances including gut dysbiosis, nutrient deficiencies, and metabolic stress. These underlying disruptions may influence immune signaling long before neurological symptoms appear.
In other words, the brain may be the site where symptoms appear, but the biological triggers can originate elsewhere.
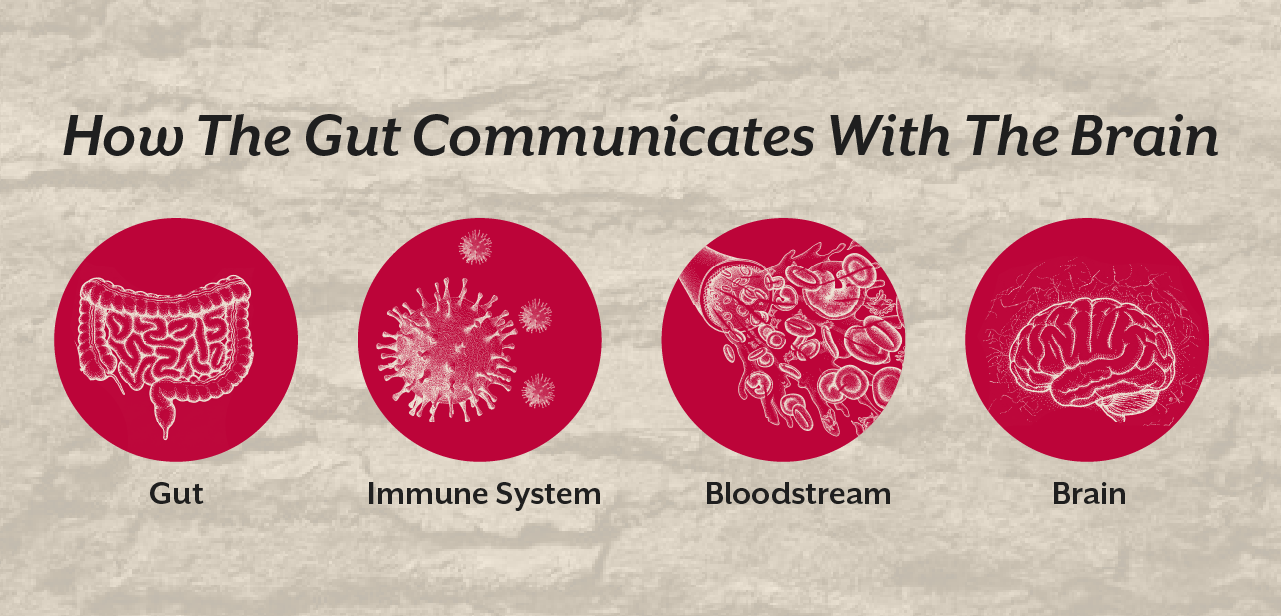
The gut brain axis describes the communication network between the digestive system and the central nervous system. This communication occurs through immune signaling molecules, the vagus nerve, microbial metabolites, and inflammatory pathways.
The gut microbiome plays an important role in regulating immune tolerance. Certain beneficial bacteria help train immune cells to recognize the difference between harmful invaders and the body’s own tissues.
When the gut microbiome becomes imbalanced, a state known as gut dysbiosis, immune regulation can weaken.
Research in the multiple sclerosis disease has identified patterns of microbiome disruption including the reduced presence of protective bacteria such as Akkermansia muciniphila. This bacterium helps maintain the protective mucosal layer of the intestine and supports intestinal barrier integrity.
When these protective organisms decline, the gut lining can become more vulnerable to inflammation and permeability changes.
This process is often referred to as intestinal permeability or leaky gut.
Once the intestinal barrier becomes compromised, microbial fragments and undigested food proteins may enter the bloodstream. These molecules can trigger immune activation and systemic inflammation that affects distant organs including the brain.
One of the most important mechanisms linking gut health to the causes of MS is intestinal permeability.
The intestinal lining normally acts as a selective barrier that allows nutrients to pass into the bloodstream while preventing toxins and pathogens from entering circulation.
In individuals with chronic inflammation, the proteins that regulate intestinal tight junctions can become disrupted. One biomarker associated with this process is zonulin.
Elevated zonulin levels are commonly observed in individuals experiencing increased intestinal permeability.
When tight junctions open excessively, foreign molecules can enter the bloodstream and stimulate the immune system. This immune activation can become chronic, creating a pro-inflammatory environment throughout the body.
Over time this inflammatory state may weaken the blood brain barrier, the protective layer that normally shields the brain from circulating immune cells.
When this barrier becomes compromised, inflammatory signals can reach the central nervous system and contribute to the demyelination process characteristic of multiple sclerosis.

Another layer of complexity in the causes of MS involves mitochondrial dysfunction.
Mitochondria are the energy producing structures inside cells that generate ATP, the molecule that powers cellular activity.
In the nervous system, neurons require enormous amounts of energy to transmit electrical signals and maintain structural integrity.
When mitochondrial function declines, neurons become more vulnerable to oxidative stress and inflammatory damage.
Studies in the MS disorder have identified markers of oxidative stress and mitochondrial impairment in both immune cells and neurons.
Elevated homocysteine levels, often associated with impaired methylation pathways, can further increase oxidative stress in the nervous system.
When energy production becomes inefficient, the nervous system struggles to repair damaged myelin and maintain normal nerve function.
This metabolic vulnerability may contribute to the progression of neurological symptoms seen in multiple sclerosis.
The composition of the gut microbiome can strongly influence immune balance.
Beneficial bacteria produce short chain fatty acids and other metabolites that help regulate inflammation and support intestinal barrier function.
When these beneficial microbes decline, harmful bacteria may gain dominance. This imbalance can amplify inflammatory signaling throughout the body.
Research in individuals with the multiple sclerosis disease has identified increased levels of certain inflammatory bacterial species along with reduced microbial diversity.
The absence of protective microbes such as Akkermansia muciniphila may weaken the gut barrier while allowing inflammatory signals to travel through the vagus nerve toward the brain.
This microbial imbalance creates a persistent loop of inflammation between the gut and the nervous system.
One of the most confusing aspects of MS disease is the delay between biological dysfunction and visible symptoms.
Gut barrier disruption, microbiome imbalance, and metabolic stress may develop gradually over many years. During this time the immune system remains chronically activated even before neurological damage becomes obvious.
When the cumulative burden of inflammation and mitochondrial stress reaches a certain threshold, symptoms begin to emerge.
Fatigue, muscle weakness, and cognitive difficulties may appear slowly before a formal diagnosis of the MS medical condition is made.
Understanding this long biological timeline is essential because it suggests that the origins of the disease may precede symptoms by many years.
Conventional multiple sclerosis treatment primarily focuses on suppressing immune activity to slow disease progression.
While these therapies can reduce relapse frequency, they often do not address the metabolic and microbial drivers that may influence immune dysregulation.
A systems approach evaluates multiple physiological layers including gut health, nutrient status, mitochondrial function, and inflammatory signaling.
At iThrive Alive this broader perspective forms the foundation of the 3 months Alive program. The goal is not only symptom management but also restoration of metabolic balance across interconnected biological systems.
This approach often includes strategies that support intestinal barrier integrity, improve microbiome diversity, replenish micronutrients, and optimize cellular energy metabolism.
For individuals experiencing persistent neurological symptoms, exploring these deeper biological factors can offer valuable insights into their condition.

If you or someone you know is living with multiple sclerosis symptoms, it is important to remember that chronic illness rarely develops from a single factor.
Instead it often emerges from the interaction between immune signaling, metabolic stress, gut health, and cellular energy systems.
By understanding these deeper biological layers, individuals may gain a clearer picture of the mechanisms influencing their health.
For those seeking a deeper evaluation of their metabolic and immune status, it may be helpful to book a root cause analysis with iThrive Alive. This process looks beyond surface level symptoms to identify the drivers influencing long term health.
Multiple sclerosis is often viewed purely as a neurological condition, yet growing research suggests that its biological roots may extend far beyond the nervous system. Gut barrier dysfunction, microbial imbalance, immune activation, and mitochondrial stress can interact over many years before symptoms appear. The gut brain axis provides an important framework for understanding how disturbances in digestive health may influence neuroinflammation and immune dysregulation. By approaching the multiple sclerosis disease through a systems perspective that includes gut health, metabolism, and cellular energy production, new opportunities may emerge for supporting long term neurological resilience and improving overall quality of life.




.svg)
For many people living with fibromyalgia, the experience goes far beyond occasional pain. It often feels like the body has lost its internal energy system. Simple daily activities become exhausting, muscles feel persistently sore, and concentration becomes difficult. Many individuals describe waking up already tired, even after what should have been a full night of sleep.
This is why fibromyalgia chronic fatigue is one of the most frustrating aspects of the condition. Patients frequently report that the exhaustion feels deeper than normal tiredness. It is not the type of fatigue that disappears after rest. Instead it persists for months or years, creating a cycle of low energy, pain, and mental fog.
For decades fibromyalgia was largely misunderstood. It was often described simply as a pain disorder, or in some cases dismissed as a psychological condition. Modern research now suggests a much deeper biological story. The condition appears to involve fibromyalgia nervous system dysfunction, altered pain processing in the brain, mitochondrial energy disruption, and chronic low grade inflammation.
At iThrive Alive we frequently observe that fibromyalgia symptoms rarely begin at the level where they appear. The pain felt in muscles and joints often originates from deeper biological disturbances involving energy metabolism and neural signaling.
Understanding this hidden biology can help explain why fibromyalgia is so exhausting and why addressing root causes may improve recovery outcomes.
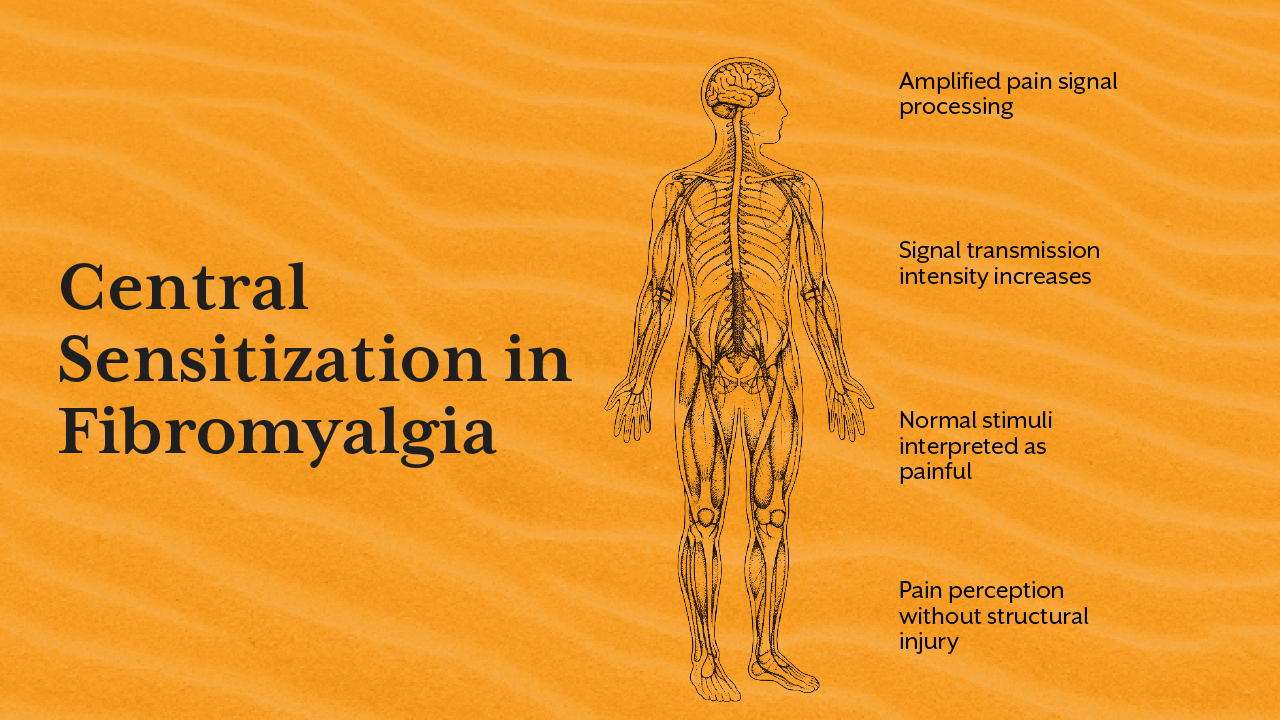
One of the most important concepts in fibromyalgia research is fibromyalgia central sensitization. This term describes a state where the nervous system becomes excessively sensitive to normal sensory signals.
In a healthy nervous system, pain signals are carefully regulated by the brain and spinal cord. However in central sensitization the brain amplifies incoming signals. Stimuli that would normally feel mild can begin to feel painful.
This helps explain why individuals with fibromyalgia may experience pain in multiple parts of the body even without clear injury. The brain essentially becomes a volume amplifier for pain signals.
Researchers have also observed that this hypersensitive state can affect other sensory experiences. Patients frequently report sensitivity to sound, light, temperature changes, and emotional stress.
This phenomenon illustrates why fibromyalgia hidden symptoms extend far beyond musculoskeletal pain. The condition involves the entire neural processing network.
Another common experience among fibromyalgia patients is cognitive impairment, often referred to as fibro fog. This symptom includes memory difficulties, reduced concentration, and slower mental processing.
Fibromyalgia brain fog causes are increasingly being linked to disruptions in brain energy metabolism. The brain consumes a large portion of the body’s total energy supply, and even small reductions in mitochondrial function can impair neural performance.
When cellular energy production declines, neurons struggle to maintain efficient communication. This may contribute to the mental fatigue that many patients describe.
Studies also show altered blood flow patterns in certain brain regions among individuals with fibromyalgia. These changes may further affect cognitive clarity and emotional regulation.
Many patients ask the same question during consultations. Why is fibromyalgia so exhausting even when physical activity is limited.
One emerging explanation involves mitochondrial dysfunction.
Mitochondria are microscopic structures within cells responsible for producing ATP, the molecule that fuels nearly every biological process. Muscles, nerves, and immune cells rely heavily on this energy supply.
When mitochondrial efficiency declines, cells struggle to maintain normal function. This creates a state of systemic energy deficit that can affect the entire body.
This biological explanation helps clarify the difference between fibromyalgia fatigue vs normal fatigue. Ordinary tiredness often results from temporary overexertion or sleep deprivation. Once rest occurs, energy levels usually return.
Fibromyalgia fatigue behaves differently. It reflects a deeper energy production problem. Even simple physical tasks may feel disproportionately draining because cells cannot generate adequate ATP.
Over time this energy shortage can contribute to chronic fatigue and pain connection seen in fibromyalgia. Muscles lacking sufficient energy may accumulate metabolic byproducts more quickly, increasing discomfort and stiffness.

Another important contributor to fibromyalgia symptoms involves chronic low grade immune activation.
Inflammatory molecules known as cytokines can influence pain perception and fatigue levels. Elevated cytokine activity has been observed in many individuals experiencing chronic fatigue conditions.
Inflammation can also interfere with mitochondrial function. Oxidative stress damages mitochondrial membranes and reduces their efficiency. This creates a feedback loop where inflammation worsens fatigue and fatigue further amplifies inflammatory signaling.
This interaction between immune activity and cellular energy metabolism may explain why fibromyalgia symptoms often fluctuate during periods of physical stress, illness, or emotional strain.
When mitochondrial output declines and nervous system sensitivity increases, the body enters a state where energy availability and pain regulation both become compromised.
Muscle tissue requires significant energy to maintain normal contraction and relaxation cycles. Without adequate ATP production, muscles remain in a partially stressed metabolic state. This can produce persistent soreness even without physical injury.
The nervous system also depends on energy intensive processes to regulate neurotransmitter balance and nerve signaling speed. When these systems slow down, pain signals may remain active longer than necessary.
This interaction explains the chronic fatigue and pain connection frequently observed in fibromyalgia.
Patients often describe feeling trapped in a cycle where fatigue limits activity, reduced activity weakens muscles, and weakened muscles further increase pain sensitivity.
At iThrive Alive we often evaluate these patterns through metabolic assessments that look beyond surface symptoms. When mitochondrial stress and inflammatory signaling are addressed together, improvements in both fatigue and pain are often observed.
Because fibromyalgia involves multiple biological systems, a narrow treatment strategy rarely produces long term improvement. At iThrive Alive the focus is often on restoring internal balance through a combination of lifestyle regulation, nutritional therapy, and targeted supplementation.
Sleep regulation is often one of the first priorities. Mitochondrial repair processes and nervous system recalibration largely occur during deep sleep cycles. Without consistent circadian rhythms the body struggles to repair damaged cellular components.
Smart eating patterns also play an important role. Stable blood glucose levels support consistent energy supply to mitochondria. Nutrient dense foods provide the micronutrients required for ATP production and antioxidant defense.
Targeted supplementation may further support mitochondrial function. Nutrients such as magnesium, B vitamins, and certain mitochondrial cofactors assist enzymes involved in energy metabolism.
Movement therapy is introduced gradually. Gentle physical activity can stimulate mitochondrial biogenesis which is the process through which cells produce new mitochondria. This helps rebuild cellular energy capacity over time.
The three month Alive program integrates these principles into a structured intervention that addresses the biological drivers of chronic fatigue rather than focusing only on symptom suppression.
For individuals who want a deeper evaluation of their underlying health patterns, the option to book a root cause analysis can help identify metabolic drivers that may be contributing to fibromyalgia symptoms.

Fibromyalgia is far more complex than a simple pain condition. It represents a biological intersection between nervous system hypersensitivity, mitochondrial energy disruption, and immune signaling imbalance. When cellular energy production declines and the nervous system becomes overly reactive, the body enters a cycle where fatigue, cognitive fog, and chronic pain reinforce each other. Understanding this hidden biology helps explain why fibromyalgia can feel so exhausting and unpredictable. At iThrive Alive the goal is not only to reduce symptoms but to restore the internal systems that regulate energy and neural communication. By focusing on mitochondrial health, lifestyle rhythms, and metabolic resilience, it becomes possible to move beyond symptom management and begin rebuilding the body’s capacity for sustained energy and recovery.




.svg)
Many people assume fatigue simply means being tired after a long day. Rest, sleep, or a short break usually restores energy. But individuals living with chronic fatigue syndrome describe something very different. Their exhaustion does not disappear with sleep. Even simple activities such as walking, working, or concentrating can feel overwhelming.
Chronic fatigue syndrome symptoms often extend far beyond tiredness. People report persistent brain fog, headaches, muscle pain, reduced stamina, and difficulty recovering after physical or mental exertion. The experience can feel confusing because medical tests frequently appear normal, leaving patients without clear explanations for their symptoms.
Increasingly, research suggests that the answer may lie deeper inside the body at the cellular level. The body’s ability to produce energy efficiently depends heavily on mitochondria, the microscopic structures inside cells that generate ATP. ATP is the biological fuel that powers muscle contraction, nerve signaling, immune regulation, and cognitive function.
When mitochondrial efficiency declines, the body struggles to maintain energy across multiple systems. What initially appears as fatigue may actually reflect widespread disruption in cellular energy production.
Understanding this biological layer changes how chronic fatigue syndrome is viewed. Instead of being dismissed as unexplained exhaustion, it becomes a condition that may involve metabolic stress, immune dysfunction, and impaired mitochondrial activity.
Chronic fatigue syndrome is sometimes referred to as chronic fatigue immune dysfunction syndrome. The name itself reflects the complexity of the condition. It does not involve only fatigue but also disturbances in immune activity, neurological signaling, and metabolic function.
Individuals experiencing chronic fatigue syndrome symptoms frequently report a cluster of overlapping issues. These may include persistent exhaustion, poor exercise tolerance, headaches, sleep disturbances, and cognitive slowing often described as brain fog.
Unlike normal fatigue, the exhaustion seen in chronic fatigue syndrome often worsens after exertion. Even mild activity can trigger prolonged fatigue lasting hours or days. This phenomenon is often referred to as post exertional malaise.
Scientists believe this pattern may occur because the body cannot produce or distribute energy efficiently during stress. Instead of generating adequate ATP, cells begin to operate under metabolic strain. As a result, the nervous system, muscles, and immune system may all struggle to maintain normal function.
This explains why chronic fatigue syndrome symptoms appear so widespread. The condition is not limited to one organ but reflects a deeper problem in cellular energy regulation.
Mitochondria are often described as the energy engines of the cell. Their primary role is to convert nutrients and oxygen into ATP through a process known as oxidative phosphorylation.
This energy is essential for nearly every biological activity including muscle movement, nerve communication, immune defense, and brain function. When mitochondrial efficiency declines, the body begins to experience a subtle but widespread energy shortage.
In individuals with chronic fatigue syndrome, researchers have observed signs of impaired mitochondrial function. Cells appear less capable of generating adequate ATP during periods of stress. As a result, the body becomes more vulnerable to fatigue, inflammation, and poor recovery after exertion.
Another important factor is oxidative stress. When mitochondria struggle to produce energy efficiently, they can generate excessive reactive oxygen species. These unstable molecules damage cellular structures and further reduce mitochondrial efficiency.
Over time, this creates a cycle in which reduced energy production leads to more oxidative stress, and oxidative stress further weakens mitochondrial performance.
This cycle may explain why fatigue in chronic fatigue syndrome feels so persistent and difficult to resolve.

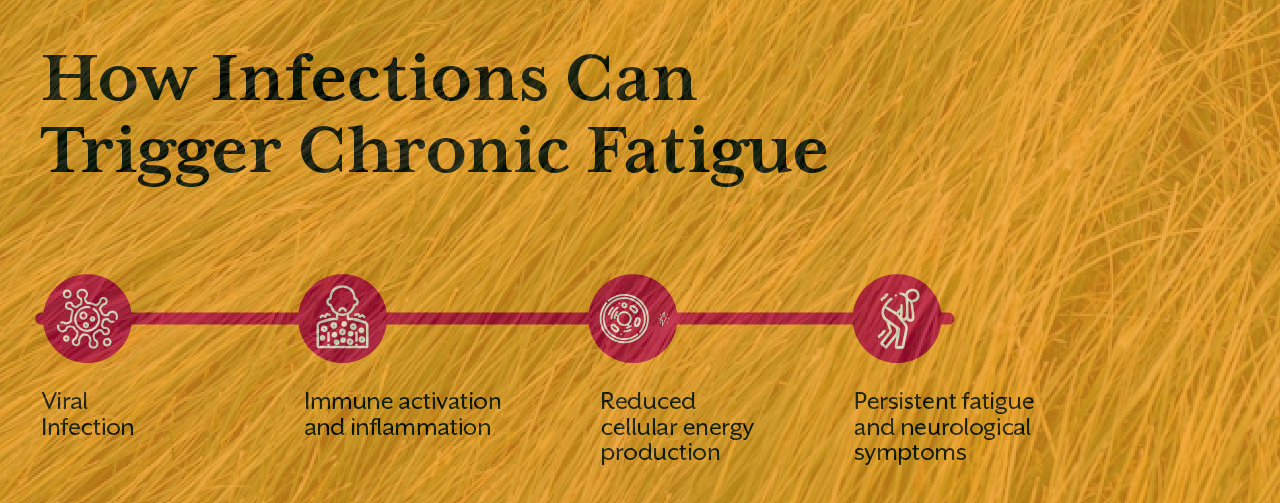
Many individuals report that their symptoms began after an infection. Viral illnesses such as influenza, Epstein Barr virus, or other infections have been linked to post viral chronic fatigue syndrome.
During an infection, the immune system activates a powerful inflammatory response to eliminate pathogens. While this response is necessary for protection, prolonged immune activation can place significant stress on cellular metabolism.
Inflammatory molecules released by immune cells can interfere with mitochondrial energy production. Instead of efficiently generating ATP, mitochondria begin to operate less effectively.
At the same time, inflammation may alter the way the brain regulates fatigue signals. The nervous system becomes more sensitive to physical stress, making even small tasks feel exhausting.
This interaction between immune activity and mitochondrial metabolism may explain why chronic fatigue immune dysfunction syndrome often develops after infections.
One of the most frustrating symptoms reported by individuals with chronic fatigue syndrome is brain fog. People describe difficulty concentrating, memory lapses, slower thinking, and mental exhaustion.
The brain consumes an enormous amount of cellular energy. Although it represents only a small percentage of body weight, it requires a constant supply of ATP to maintain neural signaling and cognitive performance.
When mitochondrial energy production declines, the brain may struggle to maintain normal function. This can lead to cognitive fatigue and difficulty processing information.
Chronic fatigue syndrome headaches may also arise from metabolic stress in the nervous system. Reduced energy availability combined with inflammation can alter pain signaling pathways, making headaches more frequent or persistent.
These neurological symptoms highlight how deeply energy metabolism influences cognitive health.
Energy production inside the body depends on more than just mitochondria. It is influenced by the broader metabolic environment including nutrition, inflammation, hormonal balance, and gut health.
Nutrient deficiencies can reduce the availability of key cofactors required for mitochondrial function. Nutrients such as magnesium, B vitamins, and CoQ10 are involved in multiple stages of ATP production.
Chronic inflammation also disrupts cellular metabolism. Inflammatory signaling diverts energy away from normal physiological functions and toward immune activation.
Gut health plays a role as well. Disturbances in the gut microbiome can influence immune regulation and nutrient absorption, both of which impact cellular energy pathways.
For individuals living with chronic fatigue syndrome, these overlapping factors can gradually reduce the body’s ability to sustain healthy energy production.
Because chronic fatigue syndrome involves multiple biological systems, addressing it often requires a broader approach than simply treating fatigue symptoms.
At iThrive Alive, the focus is on identifying the underlying drivers that may be affecting cellular energy systems. This may include evaluating nutrient status, metabolic markers, gut health, and lifestyle patterns that influence mitochondrial efficiency.
Lifestyle interventions play an important role. Regular movement can stimulate mitochondrial growth and improve metabolic flexibility. Sleep quality supports cellular repair processes that occur overnight.
Nutrition also provides essential substrates for mitochondrial function. Whole foods rich in micronutrients support enzymatic pathways involved in energy production.
Targeted supplementation may help replenish nutrients that support mitochondrial respiration and antioxidant defense.
For individuals experiencing persistent fatigue, exploring deeper metabolic patterns may offer valuable insights. Many people begin this process by choosing to book a root cause analysis through the iThrive Alive program to understand what may be influencing their symptoms.

Persistent fatigue can feel frustrating and isolating. Many individuals struggle for years without understanding why their energy levels remain low.
However, chronic fatigue syndrome symptoms may serve as an important signal from the body. When cellular energy production begins to decline, the body often reduces activity levels as a protective mechanism.
Recognizing this signal allows individuals to explore deeper biological factors that may be influencing their health. Addressing mitochondrial function, immune balance, and metabolic resilience can help restore the body’s natural energy systems over time.
For individuals looking for personalized guidance, iThrive Alive also offers the option to book a consult to explore strategies that may support energy recovery.
Chronic fatigue syndrome is far more complex than simple tiredness. The condition reflects a deeper disruption in the body’s energy producing systems. Mitochondria play a central role in this process because every biological function depends on adequate cellular energy. When mitochondrial efficiency declines due to inflammation, infections, nutrient deficiencies, or metabolic stress, the body begins to struggle with energy distribution across multiple systems. This can lead to persistent fatigue, brain fog, headaches, and poor recovery after exertion. Understanding chronic fatigue syndrome through the lens of cellular energy metabolism offers a more comprehensive way to approach the condition. By supporting mitochondrial health through nutrition, lifestyle adjustments, and targeted metabolic strategies, it may be possible to gradually restore the body’s capacity for sustainable energy.




.svg)
Most people believe disease begins when symptoms appear.
A diagnosis arrives. Pain begins. Fatigue becomes constant. Blood tests cross a threshold. Only then does the condition receive a name.
But functional nutrition tells a very different story.
Chronic illness rarely begins at the moment symptoms appear. In many cases the biological changes that eventually lead to conditions like multiple sclerosis, chronic fatigue syndrome, rheumatoid arthritis, fibromyalgia, or even metabolic disorders such as diabetes begin years earlier.
During this silent period the body is already struggling at a deeper cellular level. Energy production declines. Inflammation slowly rises. Repair mechanisms weaken. Nutrient signaling becomes inefficient. Mitochondria begin to lose efficiency.
The problem is that these early disruptions rarely show up in routine medical testing. Blood markers may still appear normal. Hormone levels may fall within acceptable ranges. Yet inside tissues, organs and cells, regulatory networks are slowly shifting toward dysfunction.
This is one of the central ideas behind the functional model used at iThrive Alive. Instead of asking where symptoms appear, we ask where the biological disruption actually began.
Very often, the answer lies in one fundamental system within the body: cellular energy production driven by mitochondria.
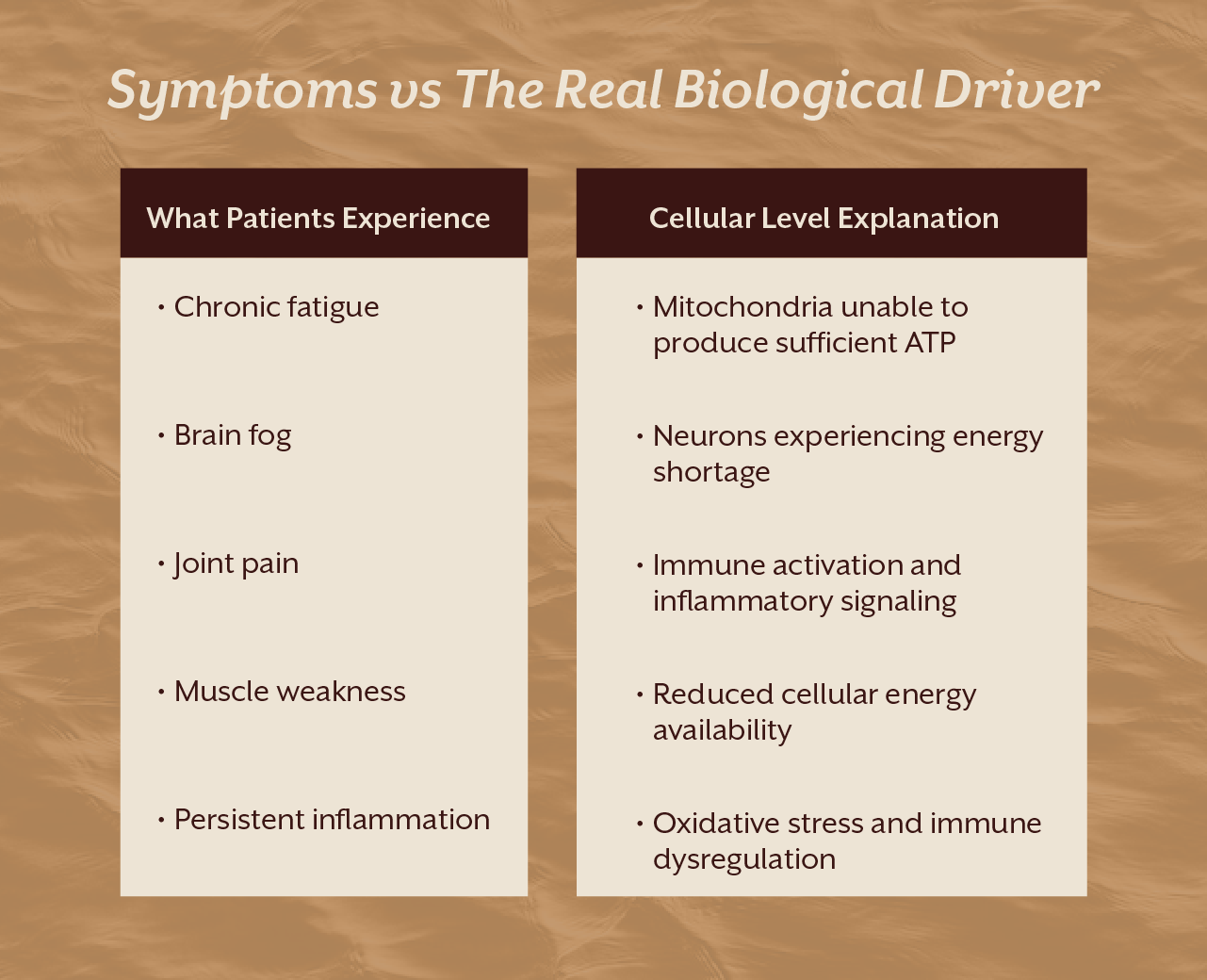
Mitochondria are often described as the energy producers of the body. But this description is only partially accurate. Their role extends far beyond energy generation. Mitochondria regulate immune signaling, control oxidative stress, influence hormone production, and determine how cells respond to metabolic signals. In many ways they function as the metabolic control centers of the cell.
When mitochondrial function begins to decline, multiple systems start to lose efficiency at the same time. Cells struggle to produce ATP. Inflammatory pathways become more active. Antioxidant defenses weaken. Tissues with high energy demand such as the brain, muscles, immune cells and endocrine glands are the first to show signs of dysfunction.
This is why conditions that appear unrelated on the surface often share a similar underlying pattern.
A person experiencing unexplained fatigue may be facing the same mitochondrial stress that later contributes to autoimmune disease. Someone struggling with insulin resistance may also experience neurological fatigue or muscle weakness because mitochondrial signaling is disrupted. From a functional nutrition perspective the disease name matters less than the underlying energy imbalance.
When mitochondrial efficiency declines, the body gradually shifts from optimal function toward survival mode. Repair slows. Recovery becomes difficult. Inflammation persists longer than it should.
Symptoms eventually emerge, but they are often only the final visible stage of a much longer process.
One of the reasons chronic illnesses appear confusing is because symptoms rarely stay limited to a single organ system.
When mitochondrial stress spreads across the body, different tissues begin responding in different ways.
Muscles may develop weakness or fatigue. The brain may struggle with concentration and cognitive clarity. The immune system may become overactive, triggering autoimmune responses. Hormonal systems may lose balance. This explains why conditions like fibromyalgia, chronic fatigue syndrome, and autoimmune disorders often involve a wide spectrum of symptoms rather than a single complaint.
For example, individuals with chronic fatigue syndrome frequently report profound exhaustion that does not improve with rest. Research suggests that their cells struggle to generate sufficient ATP despite adequate oxygen and nutrients. The energy deficit becomes systemic.
In autoimmune diseases such as multiple sclerosis or rheumatoid arthritis the situation becomes even more complex. Mitochondrial dysfunction increases oxidative stress which damages cellular structures. Damaged proteins and lipids then trigger immune responses, leading the immune system to attack tissues that appear abnormal. The symptoms appear neurological, muscular, or joint related. But the underlying disruption often originates in cellular energy metabolism. At iThrive Alive this is why symptom based treatment rarely forms the starting point of our investigation. Instead the focus shifts toward identifying deeper drivers such as mitochondrial inefficiency, gut barrier disruption, microbial imbalance, nutrient deficiencies and inflammatory load.
Only when these systems are evaluated together can the true origin of symptoms begin to emerge.
One of the reasons chronic illnesses appear confusing is because symptoms rarely stay limited to a single organ system.
When mitochondrial stress spreads across the body, different tissues begin responding in different ways.
Muscles may develop weakness or fatigue. The brain may struggle with concentration and cognitive clarity. The immune system may become overactive, triggering autoimmune responses. Hormonal systems may lose balance. This explains why conditions like fibromyalgia, chronic fatigue syndrome, and autoimmune disorders often involve a wide spectrum of symptoms rather than a single complaint.
For example, individuals with chronic fatigue syndrome frequently report profound exhaustion that does not improve with rest. Research suggests that their cells struggle to generate sufficient ATP despite adequate oxygen and nutrients. The energy deficit becomes systemic.
In autoimmune diseases such as multiple sclerosis or rheumatoid arthritis the situation becomes even more complex. Mitochondrial dysfunction increases oxidative stress which damages cellular structures. Damaged proteins and lipids then trigger immune responses, leading the immune system to attack tissues that appear abnormal. The symptoms appear neurological, muscular, or joint related. But the underlying disruption often originates in cellular energy metabolism. At iThrive Alive this is why symptom based treatment rarely forms the starting point of our investigation. Instead the focus shifts toward identifying deeper drivers such as mitochondrial inefficiency, gut barrier disruption, microbial imbalance, nutrient deficiencies and inflammatory load.
Only when these systems are evaluated together can the true origin of symptoms begin to emerge.

One of the most important ideas in metabolic medicine is that disease progression follows a timeline.
Symptoms appear only after biological stress crosses a certain threshold.
For years before this point the body attempts to compensate. Mitochondria increase their workload. Hormonal signaling adapts. The immune system adjusts its activity. These compensations can maintain stability for a surprisingly long time. But eventually the system reaches a point where compensation is no longer enough. This is when symptoms begin to surface.
Fatigue becomes persistent. Brain fog appears. Joint pain develops. Blood markers start drifting toward abnormal ranges. Sleep quality declines. Mood and cognitive clarity begin to shift.
Many people assume these changes represent the start of illness. In reality they often represent the end of a much longer biological process.
Understanding this timeline changes how chronic disease should be approached. Instead of waiting for severe symptoms or abnormal lab values, a systems model focuses on identifying the early disruptions that precede disease. Mitochondrial stress, metabolic inflexibility, gut barrier damage and chronic inflammation often appear years before diagnosis.
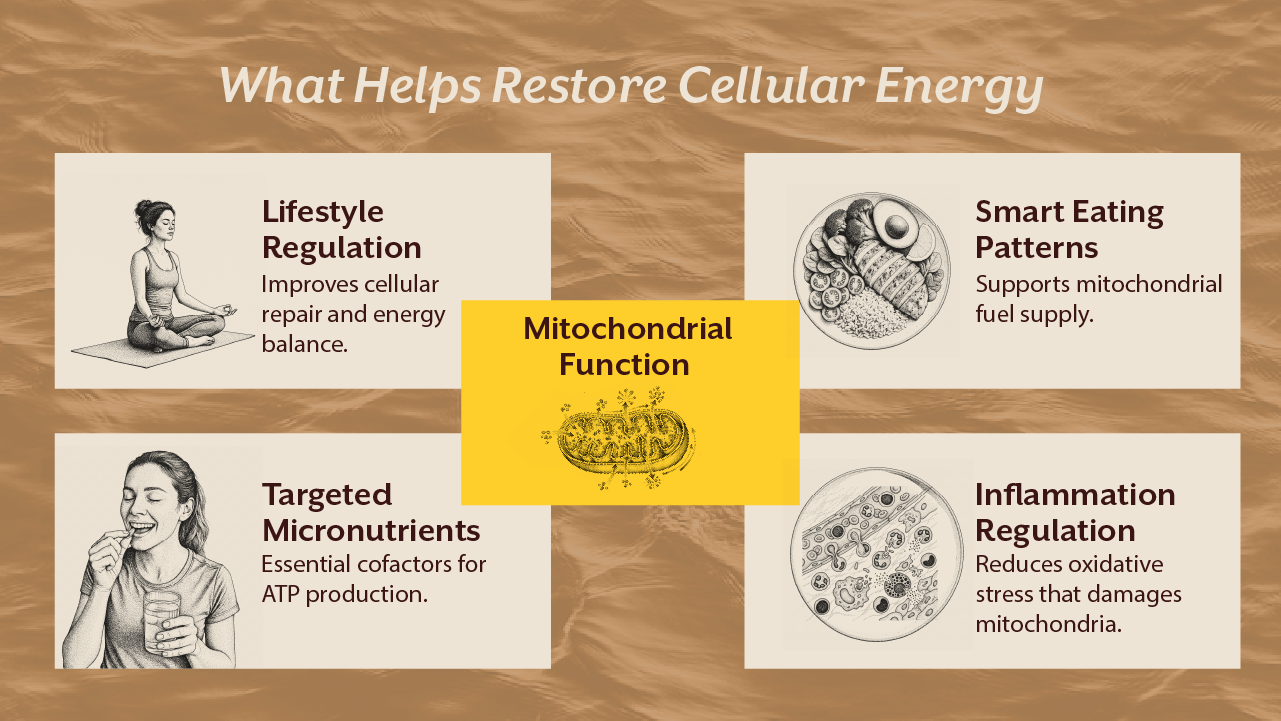
When these early patterns are addressed, the trajectory of illness can change dramatically. This is precisely the reason programs such as the iThrive Alive 3 months intervention focus heavily on restoring metabolic resilience rather than simply managing symptoms.
Lifestyle restructuring, targeted supplementation, and strategic nutritional protocols aim to restore cellular energy efficiency so the body can return to a state of repair and regulation.
Chronic illness rarely begins where symptoms appear. Conditions such as multiple sclerosis, chronic fatigue syndrome, rheumatoid arthritis and fibromyalgia often represent the final stage of a much longer biological process. Long before diagnosis the body may already be experiencing mitochondrial stress, metabolic imbalance, immune activation and reduced cellular repair capacity. Recognizing these early drivers transforms how disease should be understood and addressed. When interventions focus on restoring cellular energy systems through nutrition, lifestyle optimization and targeted supplementation, the body regains the ability to regulate inflammation, repair tissues and rebuild resilience. Chronic illness is not simply a collection of symptoms. It is a systems level disruption that begins quietly at the cellular level long before it becomes visible.




.svg)
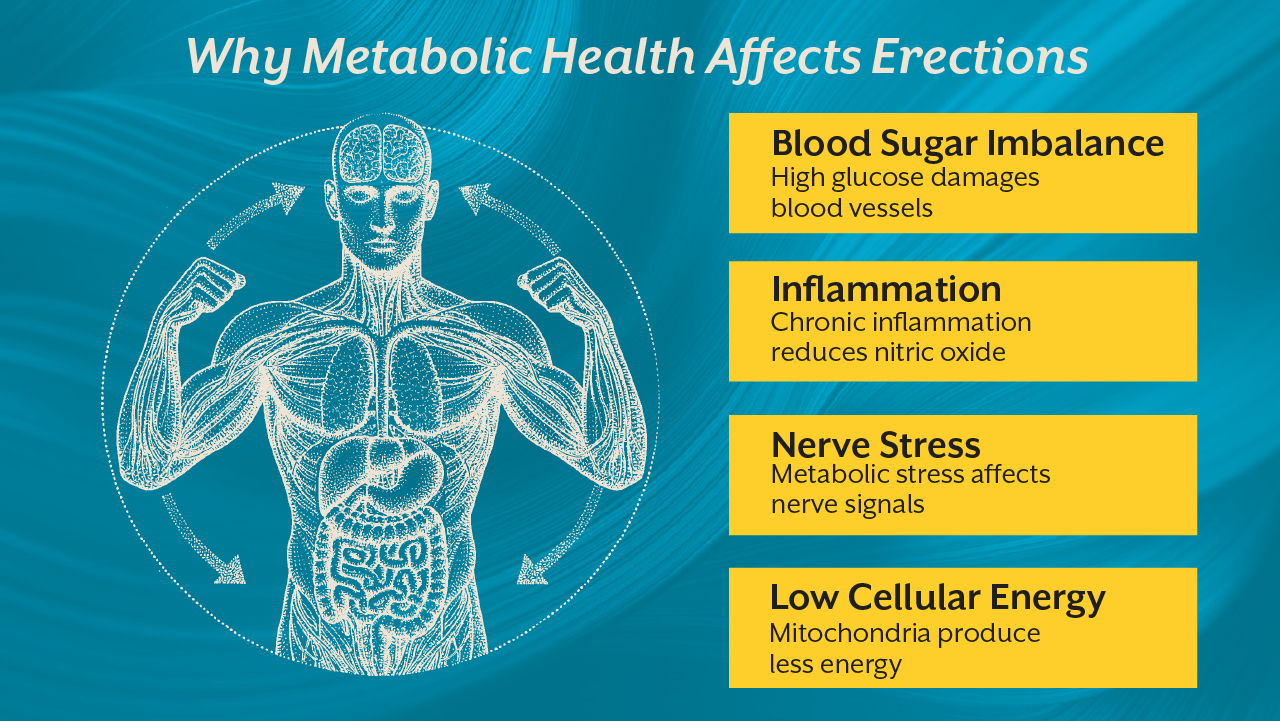
Many men silently struggle with erectile dysfunction or low sexual drive but rarely talk about it. When it does come up, the discussion often turns immediately toward medications or hormonal problems. However, there is an important biological layer that is frequently ignored. The health of the tiny energy factories inside our cells.
Erectile dysfunction, also known as impotence, is not only about hormones or aging. It is deeply connected to cellular metabolism, blood vessel function, nerve signaling, and energy production inside the body. All of these systems rely heavily on mitochondrial health.
Mitochondria are responsible for producing ATP, the energy currency that powers almost every process in the body. From muscle contraction to hormone production and blood vessel relaxation, mitochondria quietly control the physiological processes that make a healthy erection possible.
When mitochondrial function declines due to poor nutrition, chronic inflammation, metabolic stress, or insulin resistance, the body begins to lose efficiency in multiple systems. Blood vessels become less responsive, nerve signaling slows down, and hormonal balance may shift. Over time this can appear as erectile dysfunction.
For many men, erectile dysfunction is therefore not simply a sexual health issue. It may be an early metabolic signal that something deeper in the body needs attention.
Understanding this connection can completely change the way we approach erectile dysfunction and the strategies used to restore health.
Erectile dysfunction occurs when a man consistently finds it difficult to achieve or maintain an erection sufficient for sexual activity. While testosterone is often blamed for this issue, the physiology of erection is actually much more complex.
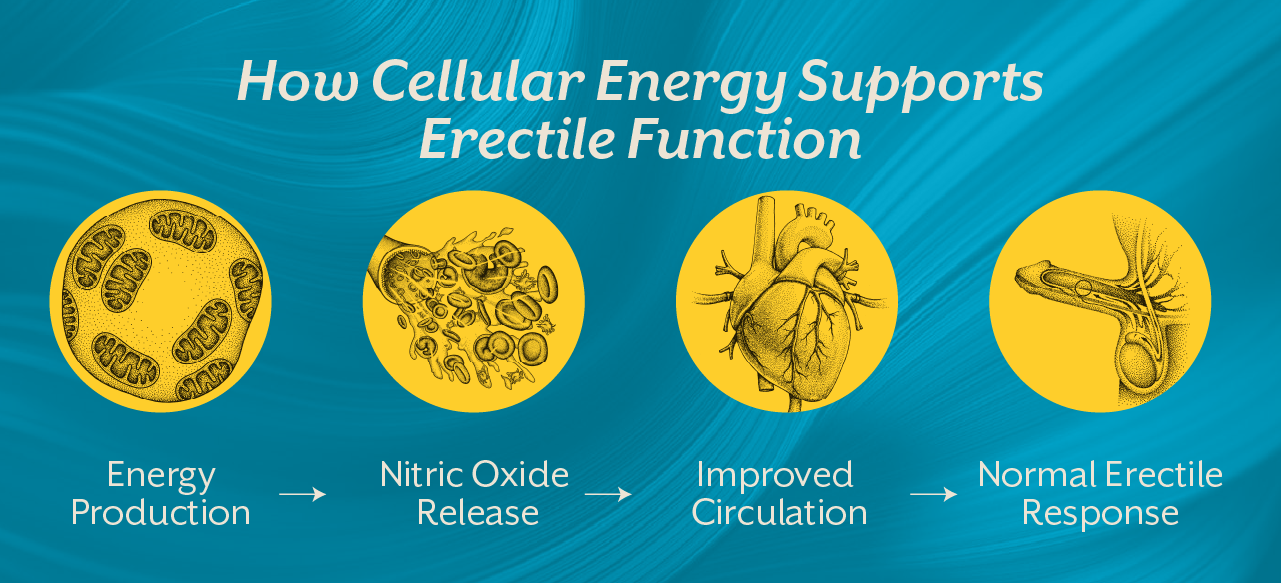
An erection depends on a coordinated interaction between blood vessels, nerve signals, hormones, and cellular metabolism. When sexual stimulation occurs, nerve signals trigger the release of nitric oxide in penile blood vessels. Nitric oxide relaxes smooth muscles and allows blood to flow into the erectile tissues.
For this process to work smoothly, blood vessels must respond properly, nerves must transmit signals efficiently, and cells must produce sufficient energy to sustain these responses.
This is where mitochondrial function becomes critically important.
Mitochondria regulate cellular energy production and help maintain the integrity of blood vessel lining known as the endothelium. When mitochondrial function is compromised, nitric oxide signaling weakens and blood vessels lose their ability to dilate effectively. The result is impaired circulation, which directly affects erectile response.
This is why erectile dysfunction is increasingly considered an early marker of metabolic and vascular health issues.
Mitochondria play a surprisingly large role in reproductive physiology. They are involved not only in energy production but also in regulating oxidative stress, hormone synthesis, and vascular health.
When mitochondrial efficiency declines, several biological disruptions begin to appear.
First, reduced ATP production limits the ability of blood vessels to relax and expand. This directly affects the blood flow required to maintain an erection.
Second, mitochondrial dysfunction increases oxidative stress. Excess oxidative stress damages endothelial cells, which are responsible for producing nitric oxide. Without adequate nitric oxide signaling, blood vessels cannot dilate effectively.
Third, mitochondria play an important role in steroid hormone metabolism. Testosterone synthesis involves several mitochondrial steps. If mitochondrial activity is impaired, hormonal balance may gradually decline.
Finally, mitochondrial dysfunction contributes to chronic inflammation and metabolic disorders. Conditions such as insulin resistance and metabolic syndrome are strongly associated with erectile dysfunction.
When viewed through this lens, erectile dysfunction becomes less about isolated reproductive organs and more about systemic metabolic health.
One of the strongest examples of metabolic influence on erectile function is seen in diabetes. Men with diabetes are significantly more likely to experience erectile dysfunction compared to the general population.
Chronic high blood sugar creates oxidative stress that damages both blood vessels and nerve fibers. This leads to endothelial dysfunction and impaired nerve signaling. Both are essential components of erectile physiology.
In addition, insulin resistance reduces mitochondrial efficiency. When cells struggle to process glucose properly, energy production becomes inefficient and inflammatory signaling increases. Over time this metabolic environment disrupts vascular responsiveness.
Many men first notice erectile changes years before diabetes is formally diagnosed. This makes erectile dysfunction an early metabolic warning sign.
If you want to understand how metabolic health influences chronic disease risk, the iThrive Alive blog titled Insulin Resistance: The Silent Phase Before Type 2 Diabetes explains how metabolic dysfunction develops long before clinical diagnosis.
The encouraging news is that mitochondrial function is highly responsive to lifestyle interventions. Unlike many structural diseases, mitochondrial efficiency can often improve when the right metabolic environment is created.
At iThrive Alive, the focus is on addressing the root drivers of dysfunction rather than only suppressing symptoms.
One of the first areas addressed is nutrition. A diet that stabilizes blood sugar and reduces inflammatory stress allows mitochondria to function more efficiently. Whole foods rich in micronutrients support enzymatic pathways involved in cellular energy production.
Smart supplementation can further assist mitochondrial function. Nutrients such as magnesium, zinc, CoQ10, and carnitine support mitochondrial respiration and antioxidant defense systems.
Lifestyle interventions also play a significant role. Regular movement improves mitochondrial density in muscle tissue and enhances insulin sensitivity. Sleep quality influences hormone regulation and mitochondrial repair processes.
Stress management is equally important because chronic stress hormones impair mitochondrial metabolism and contribute to systemic inflammation.
These changes may appear simple, but their cumulative impact on metabolic health can be profound.

Conventional management of erectile dysfunction often focuses on medications that temporarily improve blood flow. While these medications can be helpful for symptom relief, they do not address the metabolic environment that created the problem.
A root cause approach looks deeper. It examines factors such as insulin resistance, inflammation, mitochondrial health, hormonal balance, and nutrient status.
At iThrive Alive, this is where personalized metabolic assessment becomes valuable. Through detailed evaluation and targeted interventions, the goal is to identify the biological drivers behind symptoms and support long term physiological recovery.
For individuals experiencing persistent erectile dysfunction or unexplained low drive, it may be helpful to book a root cause analysis to understand the metabolic factors involved.
Many men hesitate to discuss erectile dysfunction because it feels personal and uncomfortable. Yet from a biological perspective, the body is often sending an early signal that metabolic health requires attention.
Blood vessels, hormones, and nerves are deeply connected to overall metabolic function. When these systems begin to show strain, erectile changes may appear long before other symptoms become obvious.
Rather than viewing erectile dysfunction as an isolated problem, it can be helpful to see it as an opportunity to improve long term health.
If you are experiencing these symptoms and want guidance, you can also book a consult with the nutritionist to explore personalized strategies for improving metabolic and reproductive health.
Erectile dysfunction is rarely just about sexual health. It reflects a complex interaction between vascular function, nerve signaling, hormonal balance, and cellular energy metabolism. Mitochondrial dysfunction sits at the center of this network because every step of erectile physiology depends on adequate cellular energy production. When mitochondria struggle due to poor nutrition, metabolic stress, or inflammation, the systems responsible for erectile function begin to lose efficiency. By focusing on metabolic health through nutrition, lifestyle changes, and targeted interventions, it is possible to support mitochondrial recovery and improve overall physiological resilience. Addressing the root cause not only supports reproductive health but also strengthens long term metabolic wellbeing.




.svg)
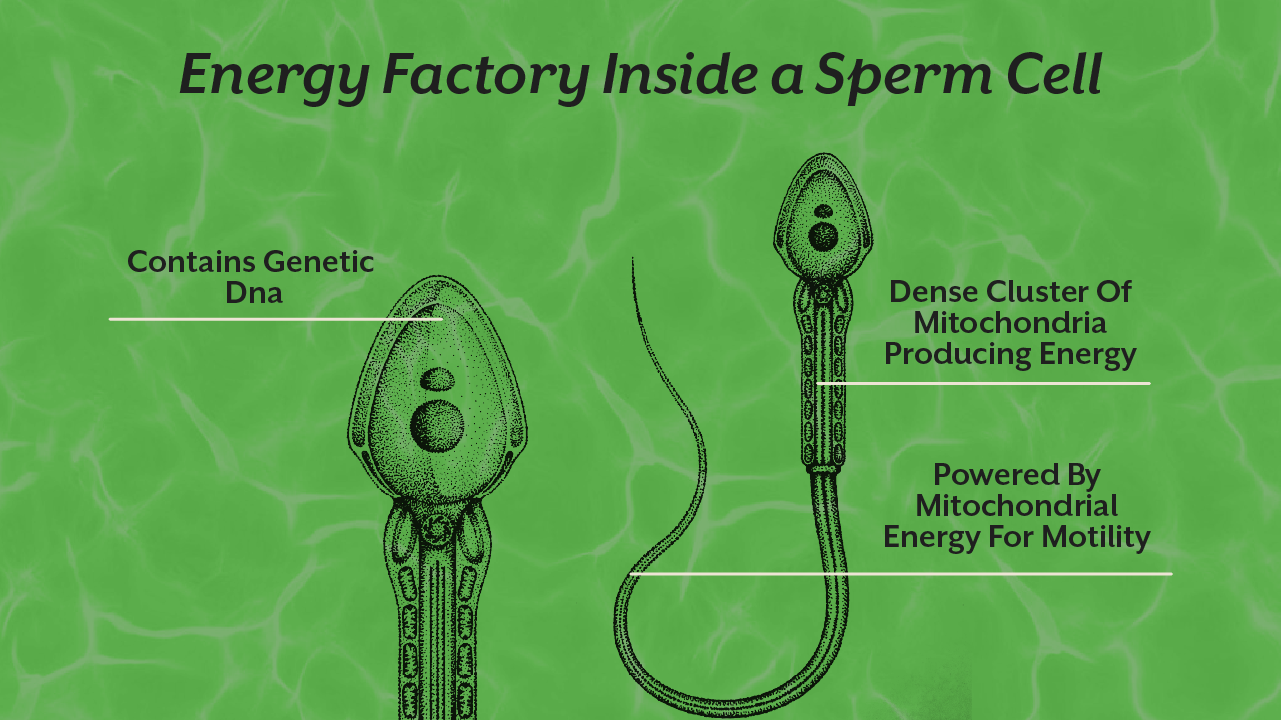
Male infertility is often approached as a reproductive problem, but biology tells a very different story. Sperm cells are among the most metabolically active cells in the human body. Their ability to move, survive, and fertilize an egg depends heavily on energy production, cellular protection, and genetic integrity. At the center of all these functions lies mitochondrial health.
Mitochondria are responsible for generating the energy required for sperm motility and survival. When mitochondrial function declines, sperm cells lose their ability to swim efficiently, maintain structural stability, and protect their DNA from oxidative damage. Over time this can manifest as low sperm count, poor motility, abnormal morphology, or in more severe cases conditions such as azoospermia.
This is one reason why many men with normal looking reports still struggle with fertility. A basic male fertility test may count sperm numbers, but it often does not evaluate cellular energy, oxidative stress, or mitochondrial efficiency. From a functional nutrition perspective these deeper metabolic layers frequently reveal the real drivers behind male infertility.
At iThrive Alive, fertility is not viewed in isolation. The clinical approach evaluates metabolic health, mitochondrial function, micronutrient status, inflammatory load, and endocrine signaling. Lifestyle interventions, targeted nutrition, and smart supplementation are then used to restore the internal environment required for healthy sperm production.
Before discussing the five nutrients that support this process, it is important to understand why mitochondrial health is central to male reproductive biology.
Sperm cells require enormous amounts of energy to travel through the reproductive tract and reach the egg. This movement, known as motility, is powered almost entirely by mitochondria located in the midpiece of the sperm cell. When mitochondrial function is compromised, the flagella lose the energy required for propulsion.
Mitochondrial dysfunction also increases the production of reactive oxygen species. In controlled amounts these molecules are part of normal cellular signaling. However excessive oxidative stress damages sperm membranes and fragments sperm DNA. Over time this reduces fertility potential even when sperm counts appear normal.
Many men experiencing male infertility show signs of metabolic stress. Blood sugar dysregulation, chronic inflammation, micronutrient depletion, and environmental toxin exposure all influence mitochondrial performance. This is why conditions such as diabetes, metabolic syndrome, and obesity are increasingly associated with reproductive challenges.
At iThrive Alive, the goal is not simply to increase sperm numbers but to improve the biological environment in which sperm are produced. Nutritional interventions that support mitochondrial function can significantly influence sperm quality.
A conventional male fertility test primarily evaluates sperm concentration, motility, and morphology. While these parameters provide useful information, they rarely explain why abnormalities occur.
For example, azoospermia may indicate the absence of sperm in semen, but the underlying causes can vary widely. Hormonal disruption, mitochondrial dysfunction, inflammatory damage, or nutrient deficiencies may all contribute.
Similarly men with borderline sperm counts may still experience infertility because their sperm cells lack adequate energy production or DNA stability. These aspects are not always visible in routine testing.
This gap between clinical symptoms and laboratory results often leaves couples searching for answers. A systems based evaluation that examines metabolic and nutritional factors can help uncover root drivers.
The iThrive Alive model integrates nutritional assessment, metabolic markers, lifestyle patterns, and functional physiology. This allows clinicians to identify factors that influence reproductive biology long before severe dysfunction develops.
Readers interested in understanding how metabolic health influences reproductive outcomes may also find insights in the iThrive Alive case study titled Targeted Nutritional Intervention in PCOS Associated Metabolic Dysfunction: A Root Cause Analysis & Functional Nutrition Driven Case Study, which explores how systemic metabolic stress influences reproductive physiology.

Zinc plays a fundamental role in testosterone production, sperm development, and antioxidant protection. It is also involved in DNA synthesis and cellular repair processes.
Low zinc status has been associated with reduced sperm count and impaired sperm morphology. Zinc also supports the activity of antioxidant enzymes that protect sperm cells from oxidative damage.
Interestingly zinc is also widely discussed in metabolic and reproductive conditions such as PCOS, which reflects how micronutrient balance influences hormonal health across genders.
Coenzyme Q10 is a key component of the mitochondrial electron transport chain. It supports cellular energy generation and acts as a powerful antioxidant within mitochondrial membranes.
Clinical studies have shown improvements in sperm motility and concentration when CoQ10 levels are optimized. Since sperm cells rely heavily on mitochondrial energy production, CoQ10 plays a central role in fertility physiology.
L Carnitine transports fatty acids into mitochondria where they are converted into energy. This process is essential for sperm motility because sperm cells depend on efficient energy metabolism for movement.
Lower levels of carnitine have been linked to reduced sperm motility. Nutritional strategies that restore carnitine levels can support cellular energy production in reproductive tissues.
Selenium functions as a cofactor for antioxidant enzymes that protect sperm cells from oxidative damage. It also contributes to thyroid hormone metabolism which indirectly influences reproductive health.
Adequate selenium intake supports sperm structural integrity and DNA stability.
Sperm membranes are rich in polyunsaturated fatty acids. Omega 3 fatty acids help maintain membrane fluidity which influences sperm motility and fertilization capability.
They also reduce inflammatory signaling which may otherwise impair reproductive function.
Readers interested in understanding how metabolic health influences reproductive physiology can also explore the iThrive Alive article What Most Nutrition Courses Get Wrong About PCOS, which explains the systemic nature of hormonal dysfunction.

While these nutrients support reproductive physiology, fertility is influenced by a broader metabolic landscape. Blood sugar regulation, gut health, inflammatory signaling, sleep patterns, and environmental exposures all affect mitochondrial efficiency.
This is why the iThrive Alive approach focuses on integrated lifestyle interventions. Smart eating strategies stabilize metabolic signaling. Targeted supplementation restores micronutrient sufficiency. Stress management improves hormonal balance. Movement improves insulin sensitivity and cellular energy production.
Rather than treating male infertility as an isolated reproductive issue, the focus remains on restoring systemic metabolic health.
Men experiencing persistent fertility challenges often benefit from deeper investigation into metabolic and nutritional drivers. Individuals seeking this level of evaluation can book a root cause analysis or book a consult with the clinical team at iThrive Alive to explore the physiological factors influencing reproductive health.
Male infertility is rarely a single organ problem. Sperm cells rely on efficient energy production, antioxidant protection, and balanced metabolic signaling. When mitochondrial function declines, sperm quality often declines with it. Nutrients such as zinc, Coenzyme Q10, L Carnitine, selenium, and omega three fatty acids support key aspects of cellular energy metabolism and reproductive physiology. However long term improvement usually requires a broader systems approach that addresses metabolic health, inflammation, lifestyle factors, and micronutrient status together. Understanding and correcting these root drivers can significantly improve the biological environment required for healthy sperm production.




.svg)
Erectile dysfunction is often discussed in whispers. Many men experience it. Few talk about it openly. A weak erection, reduced stamina, or declining sexual drive is commonly attributed to age, stress, or performance anxiety. Medication is often prescribed quickly. Yet what if impotence is not primarily a performance problem but a cellular energy problem?
At iThrive Alive, we view erectile dysfunction as a vascular and metabolic signal. An erection is not simply a mechanical event. It is a complex neurovascular response that depends on nitric oxide release, endothelial integrity, mitochondrial ATP production, and balanced hormonal signaling. When these systems are impaired, dysfunction appears.
Before labeling it psychological or inevitable, it is important to ask a deeper question. Is your body metabolically capable of sustaining optimal blood flow and cellular energy? Because in many men, the real cause of ED lies in mitochondrial dysfunction long before cardiovascular disease becomes visible.
An erection requires coordinated communication between the brain, nerves, blood vessels, and smooth muscle tissue. Sexual stimulation triggers nitric oxide release from endothelial cells lining penile arteries. Nitric oxide activates cyclic GMP pathways, relaxing smooth muscle and allowing increased blood flow into the corpus cavernosum.
This process is energy dependent. Nitric oxide synthesis requires functional endothelial cells. Endothelial cells rely heavily on mitochondria for ATP production. If mitochondrial efficiency declines, nitric oxide signaling weakens. Blood flow becomes compromised. The erection is softer or unsustained.
Mitochondrial dysfunction also increases oxidative stress. Reactive oxygen species degrade nitric oxide and damage vascular lining. Over time, this reduces vascular elasticity and responsiveness.
This is why erectile dysfunction is sometimes described as an early warning sign of cardiovascular dysfunction. The penile arteries are smaller than coronary arteries. They show dysfunction earlier.
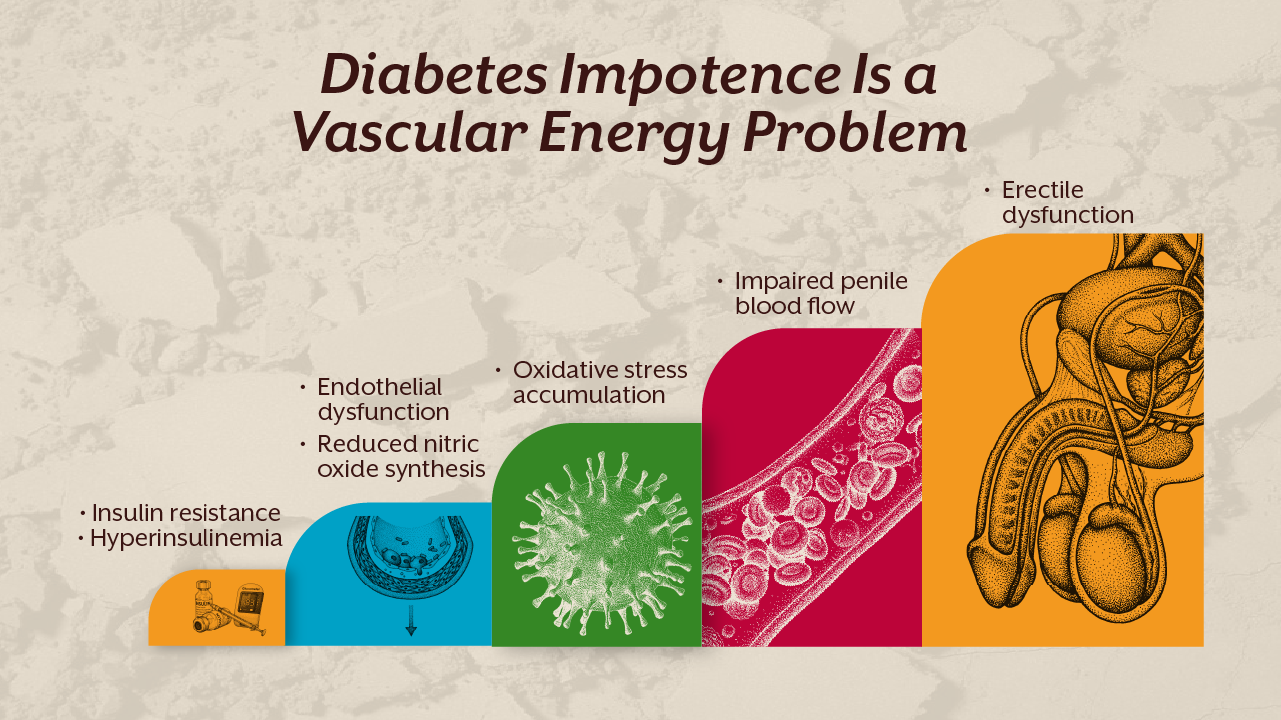
One of the strongest associations with erectile dysfunction is diabetes. In fact, diabetes impotence is not rare. Chronic hyperglycemia damages endothelial cells through advanced glycation end products. These compounds impair nitric oxide synthesis and increase oxidative stress.
But the process starts earlier than diabetes diagnosis. As discussed in our blog “Insulin Resistance: The Silent Phase Before Type 2 Diabetes”, insulin resistance precedes overt hyperglycemia by years. Elevated insulin levels impair endothelial function even before glucose rises significantly.
Hyperinsulinemia reduces nitric oxide availability. It also promotes inflammation and vascular stiffness. Over time, microvascular circulation declines. Erectile dysfunction becomes one of the earliest visible manifestations of metabolic syndrome.
Mitochondria are particularly sensitive to glucose variability. Frequent spikes in blood sugar increase reactive oxygen species production inside mitochondria. This further damages endothelial cells.
If the cause of ED is metabolic, then addressing insulin resistance is not optional. It is foundational.
Low drive is often blamed on low testosterone. While hormonal decline can contribute, testosterone production itself is mitochondria dependent. The first step of steroidogenesis occurs in the mitochondria of Leydig cells. Cholesterol conversion to pregnenolone requires mitochondrial enzymes and adequate ATP.
Chronic metabolic stress impairs this pathway. Inflammatory cytokines suppress hypothalamic signaling. Sleep deprivation reduces luteinizing hormone pulses. Micronutrient deficiencies such as zinc and magnesium impair enzymatic reactions involved in testosterone synthesis.
Therefore, libido decline is not only a hormonal issue. It is a metabolic resilience issue.
In our clinical framework at iThrive Alive, hormonal evaluation is integrated with metabolic assessment. We do not treat numbers in isolation. We assess insulin markers, inflammatory load, nutrient sufficiency, thyroid balance, and stress physiology before concluding that testosterone replacement is necessary.

If erectile dysfunction is rooted in mitochondrial dysfunction, the strategy must address cellular energy.
First, stabilize glucose variability. Smart eating types that reduce post meal spikes support mitochondrial integrity. Protein anchored meals improve satiety and reduce insulin surges. This approach aligns with principles outlined in our case study “Targeted Nutritional Intervention in PCOS Associated Metabolic Dysfunction A Root Cause Analysis and Functional Nutrition driven Case Study”, where metabolic correction restored hormonal rhythm.
Second, incorporate structured resistance training. Skeletal muscle acts as a glucose sink. Improving GLUT4 translocation enhances insulin sensitivity independent of insulin levels. Better insulin sensitivity reduces endothelial stress.
Third, prioritize micronutrient sufficiency. Magnesium supports ATP production. Zinc supports testosterone synthesis. Selenium influences thyroid mediated metabolic rate. CoQ10 supports mitochondrial electron transport chain efficiency. Supplementation should be individualized based on assessment.
Fourth, regulate sleep and circadian rhythm. Testosterone peaks during deep sleep. Mitochondrial repair processes are circadian regulated. Chronic sleep deprivation perpetuates dysfunction.
Fifth, evaluate inflammatory markers and gut health where indicated. Chronic systemic inflammation impairs vascular elasticity.
This is where a structured evaluation becomes valuable. Instead of guessing the cause of ED, men can book a root cause analysis to identify whether insulin resistance, micronutrient depletion, or inflammatory stress is the dominant driver. If you are unsure where to begin, you can book a consult to understand your metabolic landscape.

Erectile dysfunction is rarely isolated. It is often an early vascular alarm. The same metabolic pathways that lead to diabetes and cardiovascular disease influence penile blood flow. Addressing dysfunction at the mitochondrial and endothelial level offers broader protection.
Medication may improve short term blood flow. But without improving cellular energy and metabolic signaling, the underlying cause remains active.
At iThrive Alive, we focus on lifestyle intervention, smart supplementation, and precision nutrition because vascular health reflects metabolic health. When mitochondria function efficiently, endothelial cells respond effectively, hormonal rhythm stabilizes, and performance improves organically.
If you are experiencing weak erection or low drive, understand that your body may be signaling metabolic stress rather than failure. Erectile dysfunction and impotence are often early indicators of mitochondrial inefficiency, insulin resistance, and endothelial dysfunction. By restoring glucose stability, improving insulin sensitivity, supporting micronutrient status, and optimizing sleep and stress physiology, you address the biological root rather than masking the symptom. Sustainable recovery lies in cellular repair, not temporary stimulation.




.svg)
If you have been diagnosed with PCOS or polycystic ovary syndrome and are trying to conceive, the experience can feel confusing and emotionally exhausting. You may have been told that ovulation is irregular, that your hormones are imbalanced, or that you might need assisted reproductive procedures. What is often not explained clearly is why PCOS affects fertility in the first place and what you can actually do to improve your chances of pregnancy.
At iThrive Alive, we view PCOS not as a reproductive disorder alone, but as a systemic metabolic and inflammatory condition that alters ovarian signaling. PCOS pregnancy challenges are rarely about the ovaries in isolation. They are usually about insulin resistance, mitochondrial dysfunction, chronic inflammation, micronutrient depletion, and stress physiology altering how the ovaries respond to hormonal signals.
The good news is this. When the internal environment improves, ovulation improves. When metabolic signaling improves, fertility improves. And when root causes are addressed, the need for aggressive interventions often reduces.
Let us understand how.
PCOS fertility treatment is often approached as an ovulation problem. However, ovulation is only the final event in a long cascade of metabolic and hormonal signaling. In polycystic ovary syndrome, the disruption begins much earlier.
Insulin resistance plays a central role. Elevated insulin levels stimulate ovarian theca cells to produce excess androgens. These androgens interfere with follicular maturation. Instead of one dominant follicle maturing and releasing an egg, multiple immature follicles remain arrested. This is why ultrasound often shows a polycystic pattern.
Mitochondrial dysfunction adds another layer. The oocyte is one of the most energy demanding cells in the human body. If cellular energy production is compromised due to oxidative stress or micronutrient insufficiency, egg quality declines even if ovulation is induced.
Chronic inflammation further disrupts hormone receptor sensitivity. Even when hormone levels appear within reference range, cellular responsiveness may be impaired.
This is why standard reports may look acceptable, yet conception does not occur.
Most women with PCOS are advised to lose weight. While weight management can help, focusing only on the scale misses deeper metabolic drivers.
Insulin resistance increases circulating insulin even before blood glucose rises. Hyperinsulinemia worsens androgen excess and impairs ovulatory rhythm. This metabolic pattern is similar to early stage metabolic dysfunction discussed in our blog “Insulin Resistance: The Silent Phase Before Type 2 Diabetes”. Fertility disruption often begins years before diabetes risk becomes visible.
Inflammation originating from gut dysbiosis or excess visceral fat further amplifies ovarian stress. The ovaries are highly sensitive to inflammatory cytokines.
Micronutrient deficiencies such as magnesium, zinc, selenium, vitamin D, and B vitamins impair hormone synthesis and detoxification pathways.
Sleep disruption and chronic stress increase cortisol, which alters GnRH pulsatility from the hypothalamus. Ovulation depends on precise hormonal timing. Chronic stress scrambles that rhythm.
When women ask about the best treatment for PCOS to get pregnant, the answer lies in restoring metabolic flexibility, improving insulin sensitivity, reducing inflammation, and supporting cellular energy.

Many women are prescribed ovulation induction medications. While these may stimulate ovulation, they do not correct underlying metabolic dysfunction. Without addressing insulin resistance and inflammation, conception may remain difficult or early miscarriage risk may increase.
At iThrive Alive, the approach begins with root cause evaluation. Fasting insulin, HOMA IR, inflammatory markers, nutrient panels, thyroid optimization, and gut health assessment help identify drivers.
Lifestyle intervention is foundational. Smart eating types that stabilize blood glucose, structured resistance training to improve insulin sensitivity, circadian aligned sleep, and nervous system regulation are non-negotiable pillars.
Targeted supplementation supports cellular repair. In specific cases, inositol for insulin signaling, omega-3 fatty acids for inflammation control, magnesium for glucose metabolism, and antioxidant support for mitochondrial resilience are integrated.
We have seen this transformation in our published case study “Targeted Nutritional Intervention in PCOS-Associated Metabolic Dysfunction: A Root Cause Analysis & Functional Nutrition driven Case Study”. When metabolic markers improved, menstrual cycles regularized and ovulation resumed naturally.

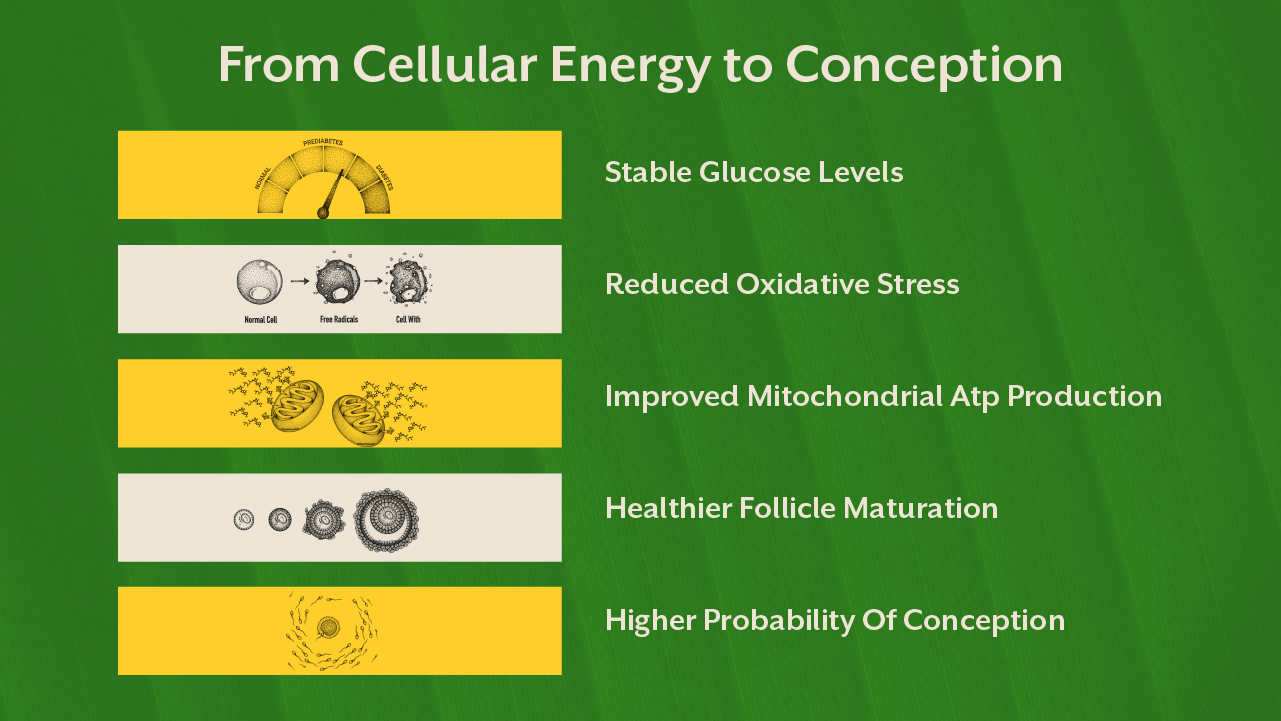
PCOS pregnancy success is not just about ovulation frequency. Egg quality matters profoundly. Mitochondria within the oocyte determine energy supply for fertilization and early embryo development.
Oxidative stress damages mitochondrial DNA. High glucose fluctuations increase free radical production. Micronutrient insufficiency reduces antioxidant defense systems.
Nutrients such as CoQ10, magnesium, B vitamins, and omega-3 fatty acids support mitochondrial efficiency. However, supplementation without dietary correction rarely produces sustainable change.
When women feel frustrated that cycles are irregular or that fertility treatments have failed, it is important to understand that improving cellular energy is a gradual biological process. Internal repair precedes visible reproductive change.
Polycystic ovary syndrome is not a barrier sentence. It is a signal. A signal that metabolic systems require attention.
When gut integrity improves, inflammation reduces. When insulin sensitivity improves, androgen excess declines. When mitochondria function efficiently, egg quality improves. When stress physiology stabilizes, ovulatory rhythm normalizes.
This is why we emphasize evaluation beyond basic hormone panels. Through a structured root cause analysis, women gain clarity about why ovulation is irregular and what is blocking conception. For those seeking guidance, you can book a root cause analysis or book a consult to understand your unique metabolic drivers.
PCOS fertility treatment is most effective when it restores biological intelligence rather than overriding it.
If pregnancy is your goal, the sequence in which you repair physiology matters. Ovulation is the last domino. Metabolic signaling is the first.
In PCOS, hyperinsulinemia stimulates ovarian theca cells to overproduce androgens via insulin receptor cross activation of LH signaling pathways. Elevated insulin also suppresses hepatic SHBG production, increasing free testosterone.
What this means practically:
Stabilize postprandial glucose excursions.
Anchor meals around adequate protein.
Avoid frequent grazing that perpetuates insulin spikes.
Incorporate resistance training to enhance GLUT4 translocation in skeletal muscle independent of insulin.
When insulin reduces, androgen excess gradually declines, allowing follicular selection to normalize.
Inflammatory cytokines such as TNF alpha and IL 6 interfere with granulosa cell function and steroidogenesis. Chronic low grade inflammation also worsens insulin resistance in a feedback loop.
Clinically, this means:
Repair gut barrier integrity.
Improve microbial diversity.
Ensure omega-3 sufficiency.
Prioritize sleep because sleep restriction elevates CRP and cortisol.
Reducing inflammatory load improves ovarian microenvironment quality.
Ovarian steroidogenesis is nutrient dependent. Magnesium acts as a cofactor in ATP dependent enzymatic reactions. Zinc supports follicular development. Selenium influences thyroid conversion which indirectly affects ovulatory rhythm. B vitamins support methylation pathways essential for hormone detoxification.
Without micronutrient sufficiency, even pharmacological ovulation induction may yield suboptimal egg quality.
Testing and repletion must be personalized rather than supplemented blindly.
The oocyte contains the highest mitochondrial density of any cell in the female body. Mitochondrial dysfunction reduces ATP availability required for meiotic spindle formation and embryo development.
Interventions include:
Improving glycemic variability.
Correcting oxidative stress.
Targeted antioxidants such as CoQ10 where clinically indicated.
Strength training to improve systemic mitochondrial biogenesis.
Circadian alignment to enhance cellular repair cycles.
Egg quality restoration is gradual because folliculogenesis spans several months.
Chronic sympathetic dominance alters hypothalamic GnRH pulse frequency. This disrupts LH and FSH balance, further impairing ovulation.
Nervous system regulation, breath work, light exposure, consistent sleep timing, and emotional stress processing are not secondary. They directly influence reproductive hormone rhythm.
Before proceeding to aggressive assisted fertility protocols, evaluate:
This is where a structured root cause analysis becomes valuable. It prevents guesswork and reduces unnecessary hormonal exposure.
For women who want clarity instead of assumptions, you can book a root cause analysis or book a consult to understand which layer of dysfunction is most relevant in your case.
If you are navigating PCOS and longing for pregnancy, understand that your body is not broken. It is responding to metabolic signals that can be influenced. Ovulation is a reflection of systemic health. By addressing insulin resistance, inflammation, mitochondrial dysfunction, and lifestyle rhythms, fertility potential improves organically. The best treatment for PCOS to get pregnant is not a single pill or protocol. It is a coordinated restoration of metabolic balance that allows the ovaries to function as they were designed to.


A complete blood panel of 60+ parameters, collected from your home by a phlebotomist. Analysis by a dedicated functional nutritionist using optimal ranges, not the standard reference ranges your doctor uses. Your symptoms, lifestyle, and health history factored into the analysis. And a 60 to 90-minute one-on-one video consultation where your nutritionist walks you through everything they found. The blood tests are included in the price. There are no hidden charges or extra cost for anything.

A complete blood panel of 60+ parameters, collected from your home by a phlebotomist. Analysis by a dedicated functional nutritionist using optimal ranges, not the standard reference ranges your doctor uses. Your symptoms, lifestyle, and health history factored into the analysis. And a 60 to 90-minute one-on-one video consultation where your nutritionist walks you through everything they found. The blood tests are included in the price. There are no hidden charges or extra cost for anything.

A complete blood panel of 60+ parameters, collected from your home by a phlebotomist. Analysis by a dedicated functional nutritionist using optimal ranges, not the standard reference ranges your doctor uses. Your symptoms, lifestyle, and health history factored into the analysis. And a 60 to 90-minute one-on-one video consultation where your nutritionist walks you through everything they found. The blood tests are included in the price. There are no hidden charges or extra cost for anything.

A complete blood panel of 60+ parameters, collected from your home by a phlebotomist. Analysis by a dedicated functional nutritionist using optimal ranges, not the standard reference ranges your doctor uses. Your symptoms, lifestyle, and health history factored into the analysis. And a 60 to 90-minute one-on-one video consultation where your nutritionist walks you through everything they found. The blood tests are included in the price. There are no hidden charges or extra cost for anything.

A complete blood panel of 60+ parameters, collected from your home by a phlebotomist. Analysis by a dedicated functional nutritionist using optimal ranges, not the standard reference ranges your doctor uses. Your symptoms, lifestyle, and health history factored into the analysis. And a 60 to 90-minute one-on-one video consultation where your nutritionist walks you through everything they found. The blood tests are included in the price. There are no hidden charges or extra cost for anything.

A complete blood panel of 60+ parameters, collected from your home by a phlebotomist. Analysis by a dedicated functional nutritionist using optimal ranges, not the standard reference ranges your doctor uses. Your symptoms, lifestyle, and health history factored into the analysis. And a 60 to 90-minute one-on-one video consultation where your nutritionist walks you through everything they found. The blood tests are included in the price. There are no hidden charges or extra cost for anything.

A complete blood panel of 60+ parameters, collected from your home by a phlebotomist. Analysis by a dedicated functional nutritionist using optimal ranges, not the standard reference ranges your doctor uses. Your symptoms, lifestyle, and health history factored into the analysis. And a 60 to 90-minute one-on-one video consultation where your nutritionist walks you through everything they found. The blood tests are included in the price. There are no hidden charges or extra cost for anything.



