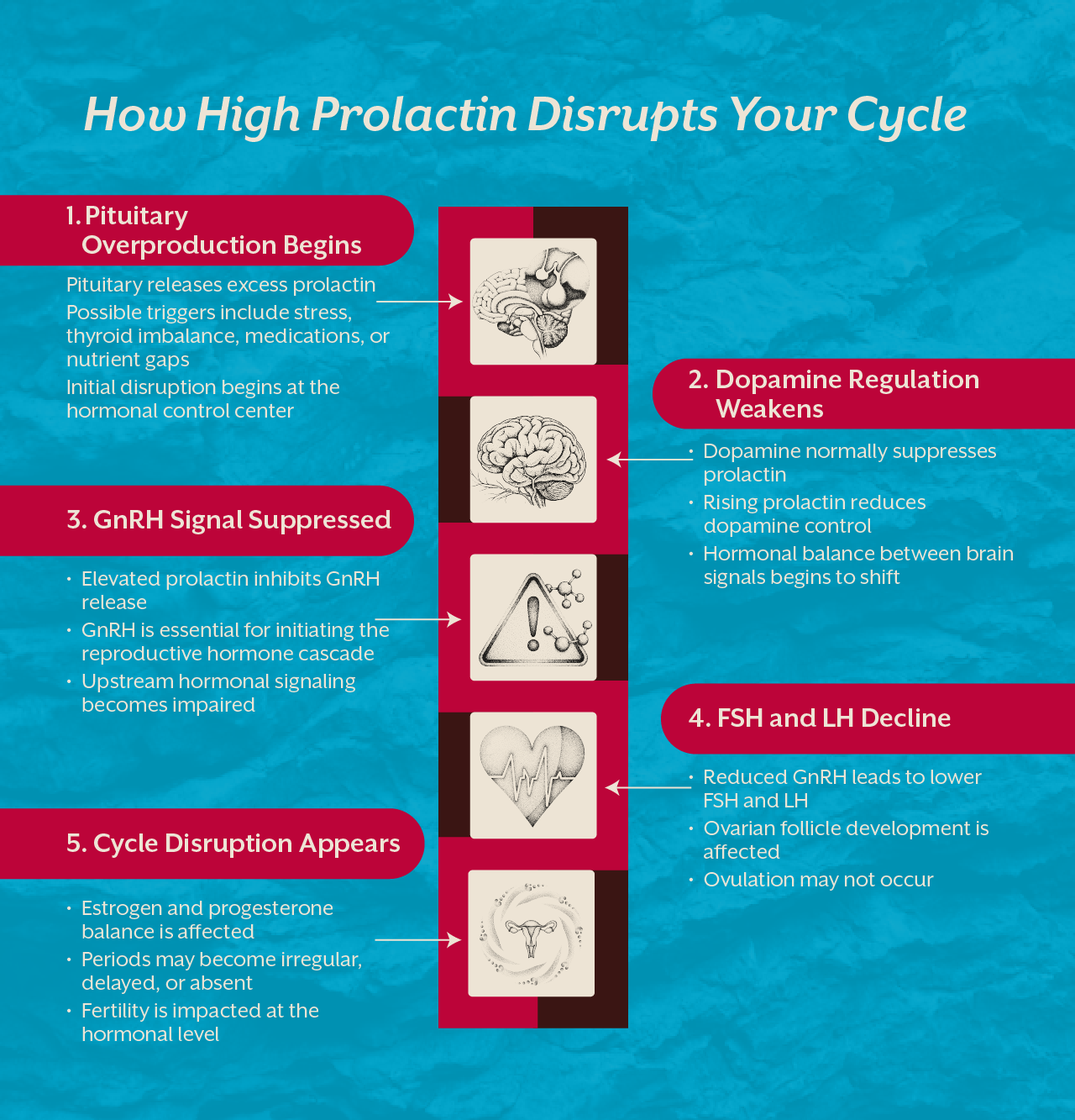
Introduction
Male infertility is often approached as a reproductive problem, but biology tells a very different story. Sperm cells are among the most metabolically active cells in the human body. Their ability to move, survive, and fertilize an egg depends heavily on energy production, cellular protection, and genetic integrity. At the center of all these functions lies mitochondrial health.
Mitochondria are responsible for generating the energy required for sperm motility and survival. When mitochondrial function declines, sperm cells lose their ability to swim efficiently, maintain structural stability, and protect their DNA from oxidative damage. Over time this can manifest as low sperm count, poor motility, abnormal morphology, or in more severe cases conditions such as azoospermia.
This is one reason why many men with normal looking reports still struggle with fertility. A basic male fertility test may count sperm numbers, but it often does not evaluate cellular energy, oxidative stress, or mitochondrial efficiency. From a functional nutrition perspective these deeper metabolic layers frequently reveal the real drivers behind male infertility.
At iThrive Alive, fertility is not viewed in isolation. The clinical approach evaluates metabolic health, mitochondrial function, micronutrient status, inflammatory load, and endocrine signaling. Lifestyle interventions, targeted nutrition, and smart supplementation are then used to restore the internal environment required for healthy sperm production.
Before discussing the five nutrients that support this process, it is important to understand why mitochondrial health is central to male reproductive biology.
Why Mitochondria Matter in Male Fertility
Sperm cells require enormous amounts of energy to travel through the reproductive tract and reach the egg. This movement, known as motility, is powered almost entirely by mitochondria located in the midpiece of the sperm cell. When mitochondrial function is compromised, the flagella lose the energy required for propulsion.
Mitochondrial dysfunction also increases the production of reactive oxygen species. In controlled amounts these molecules are part of normal cellular signaling. However excessive oxidative stress damages sperm membranes and fragments sperm DNA. Over time this reduces fertility potential even when sperm counts appear normal.
Many men experiencing male infertility show signs of metabolic stress. Blood sugar dysregulation, chronic inflammation, micronutrient depletion, and environmental toxin exposure all influence mitochondrial performance. This is why conditions such as diabetes, metabolic syndrome, and obesity are increasingly associated with reproductive challenges.
At iThrive Alive, the goal is not simply to increase sperm numbers but to improve the biological environment in which sperm are produced. Nutritional interventions that support mitochondrial function can significantly influence sperm quality.
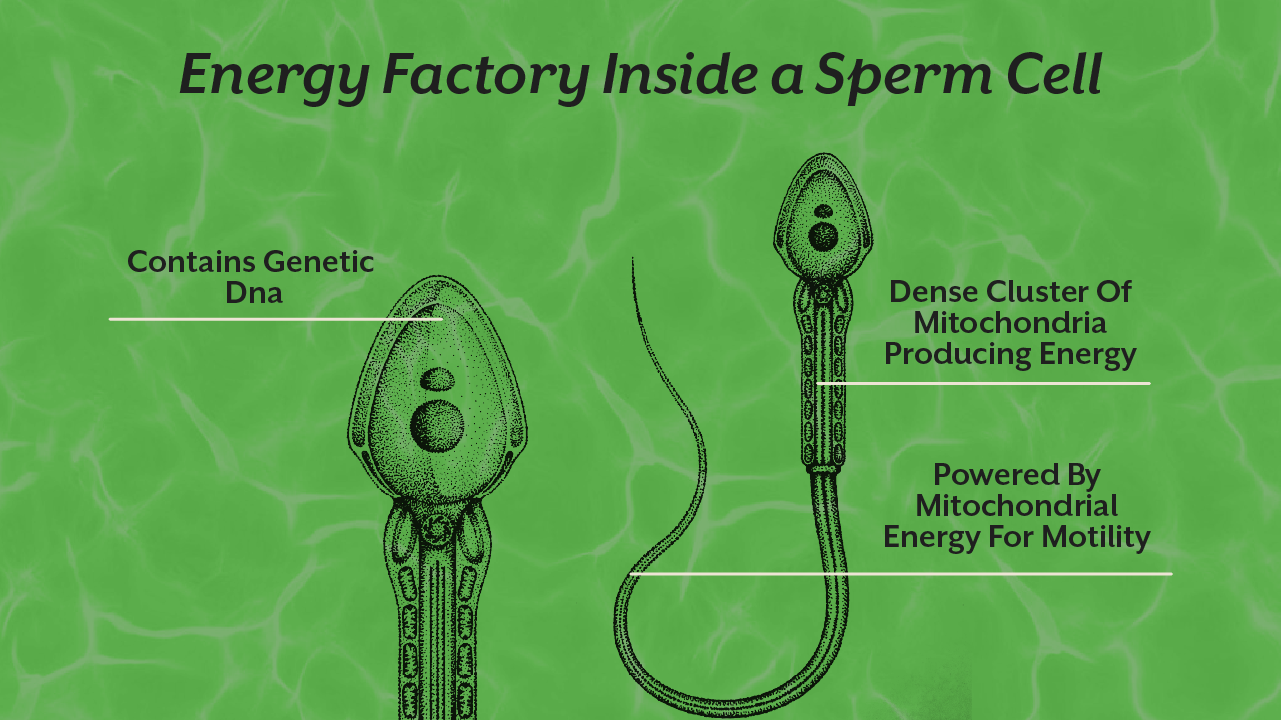
What Standard Male Fertility Tests May Miss
A conventional male fertility test primarily evaluates sperm concentration, motility, and morphology. While these parameters provide useful information, they rarely explain why abnormalities occur.
For example, azoospermia may indicate the absence of sperm in semen, but the underlying causes can vary widely. Hormonal disruption, mitochondrial dysfunction, inflammatory damage, or nutrient deficiencies may all contribute.
Similarly men with borderline sperm counts may still experience infertility because their sperm cells lack adequate energy production or DNA stability. These aspects are not always visible in routine testing.
This gap between clinical symptoms and laboratory results often leaves couples searching for answers. A systems based evaluation that examines metabolic and nutritional factors can help uncover root drivers.
The iThrive Alive model integrates nutritional assessment, metabolic markers, lifestyle patterns, and functional physiology. This allows clinicians to identify factors that influence reproductive biology long before severe dysfunction develops.
Readers interested in understanding how metabolic health influences reproductive outcomes may also find insights in the iThrive Alive case study titled Targeted Nutritional Intervention in PCOS Associated Metabolic Dysfunction: A Root Cause Analysis & Functional Nutrition Driven Case Study, which explores how systemic metabolic stress influences reproductive physiology.

Five Nutrients That Support Male Fertility
1. Zinc
Zinc plays a fundamental role in testosterone production, sperm development, and antioxidant protection. It is also involved in DNA synthesis and cellular repair processes.
Low zinc status has been associated with reduced sperm count and impaired sperm morphology. Zinc also supports the activity of antioxidant enzymes that protect sperm cells from oxidative damage.
Interestingly zinc is also widely discussed in metabolic and reproductive conditions such as PCOS, which reflects how micronutrient balance influences hormonal health across genders.
2. Coenzyme Q10
Coenzyme Q10 is a key component of the mitochondrial electron transport chain. It supports cellular energy generation and acts as a powerful antioxidant within mitochondrial membranes.
Clinical studies have shown improvements in sperm motility and concentration when CoQ10 levels are optimized. Since sperm cells rely heavily on mitochondrial energy production, CoQ10 plays a central role in fertility physiology.
3. L Carnitine
L Carnitine transports fatty acids into mitochondria where they are converted into energy. This process is essential for sperm motility because sperm cells depend on efficient energy metabolism for movement.
Lower levels of carnitine have been linked to reduced sperm motility. Nutritional strategies that restore carnitine levels can support cellular energy production in reproductive tissues.
4. Selenium
Selenium functions as a cofactor for antioxidant enzymes that protect sperm cells from oxidative damage. It also contributes to thyroid hormone metabolism which indirectly influences reproductive health.
Adequate selenium intake supports sperm structural integrity and DNA stability.
5. Omega 3 Fatty Acids
Sperm membranes are rich in polyunsaturated fatty acids. Omega 3 fatty acids help maintain membrane fluidity which influences sperm motility and fertilization capability.
They also reduce inflammatory signaling which may otherwise impair reproductive function.
Readers interested in understanding how metabolic health influences reproductive physiology can also explore the iThrive Alive article What Most Nutrition Courses Get Wrong About PCOS, which explains the systemic nature of hormonal dysfunction.

Nutrition Alone Is Not Enough
While these nutrients support reproductive physiology, fertility is influenced by a broader metabolic landscape. Blood sugar regulation, gut health, inflammatory signaling, sleep patterns, and environmental exposures all affect mitochondrial efficiency.
This is why the iThrive Alive approach focuses on integrated lifestyle interventions. Smart eating strategies stabilize metabolic signaling. Targeted supplementation restores micronutrient sufficiency. Stress management improves hormonal balance. Movement improves insulin sensitivity and cellular energy production.
Rather than treating male infertility as an isolated reproductive issue, the focus remains on restoring systemic metabolic health.
Men experiencing persistent fertility challenges often benefit from deeper investigation into metabolic and nutritional drivers. Individuals seeking this level of evaluation can book a root cause analysis or book a consult with the clinical team at iThrive Alive to explore the physiological factors influencing reproductive health.
Key Takeaway
Male infertility is rarely a single organ problem. Sperm cells rely on efficient energy production, antioxidant protection, and balanced metabolic signaling. When mitochondrial function declines, sperm quality often declines with it. Nutrients such as zinc, Coenzyme Q10, L Carnitine, selenium, and omega three fatty acids support key aspects of cellular energy metabolism and reproductive physiology. However long term improvement usually requires a broader systems approach that addresses metabolic health, inflammation, lifestyle factors, and micronutrient status together. Understanding and correcting these root drivers can significantly improve the biological environment required for healthy sperm production.


.svg)
.svg)