Introduction
When most people hear the words rheumatoid arthritis, they imagine swollen joints, stiff fingers, and persistent pain. The disease is often described as a joint condition where the immune system mistakenly attacks the lining of joints. This explanation is technically correct but biologically incomplete.
Many individuals diagnosed with rheumatoid arthritis report something else long before joint swelling appears. They experience fatigue, digestive discomfort, brain fog, poor recovery after exertion, or unexplained inflammation across the body. These symptoms rarely seem connected to arthritis at first. Yet in many cases they are the earliest signs that something deeper is happening.
Autoimmune arthritis does not begin inside the joints alone. It reflects systemic changes within the immune system, metabolism, and cellular energy networks. One emerging driver receiving growing scientific attention is mitochondrial dysfunction. Mitochondria are the cellular structures responsible for generating energy inside every cell of the body. When these energy systems begin to fail, immune regulation changes and inflammation increases.
This shift can quietly create the biological environment that later manifests as rheumatoid arthritis symptoms.
Understanding these hidden mechanisms opens a different perspective on RA treatment. Instead of focusing solely on suppressing symptoms, it becomes possible to examine the deeper biological conditions that allowed the disease to emerge.
Rheumatoid Arthritis Is a Systemic Immune Condition
Rheumatoid arthritis is classified as an autoimmune arthritis disorder in which the immune system mistakenly targets healthy tissue. The most visible target becomes the synovial membrane that surrounds joints. Inflammation within this tissue produces swelling, stiffness, and joint pain.
However the immune system does not operate in isolation from the rest of the body. Immune behavior is heavily influenced by metabolic signals, cellular energy production, and nutrient availability.
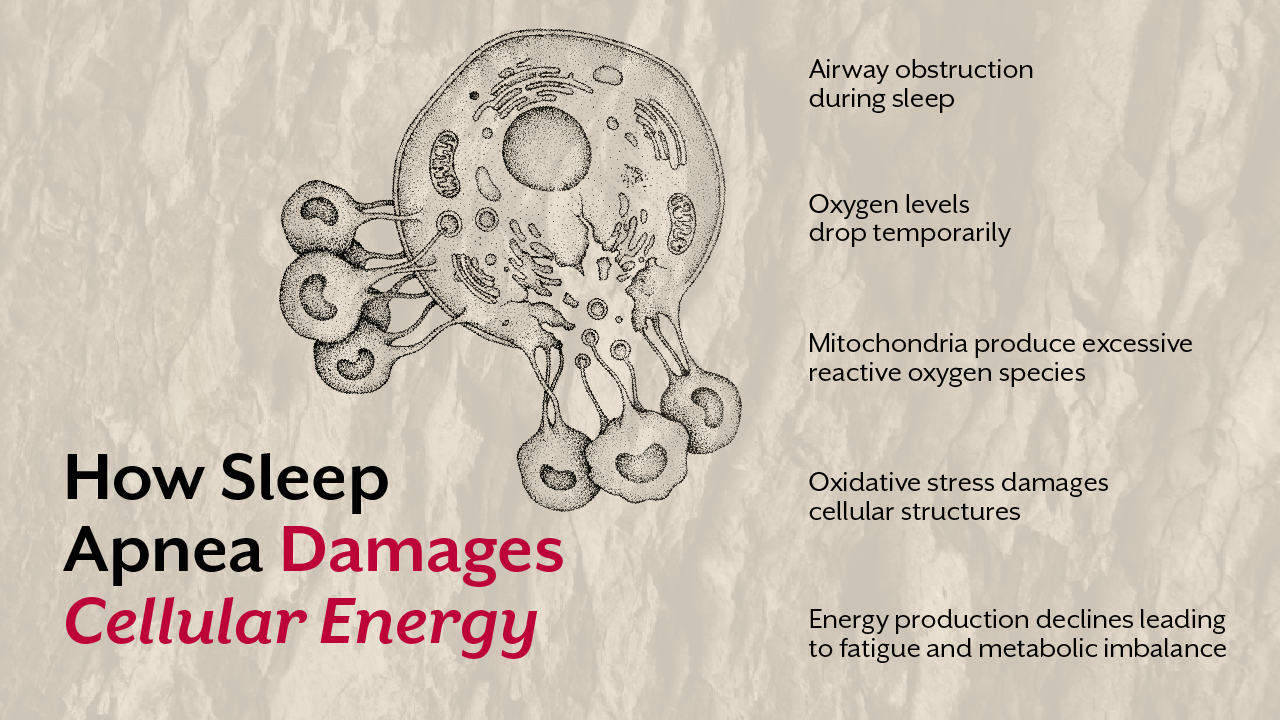
In many individuals with rheumatoid arthritis symptoms, researchers have identified disturbances in mitochondrial metabolism within immune cells. When mitochondrial energy production declines, immune cells shift toward a more inflammatory state.
This shift activates inflammatory pathways that release cytokines such as TNF alpha and interleukin six. These molecules amplify inflammation in rheumatoid arthritis and contribute to joint damage over time.
The joints therefore become the visible site of inflammation. But the origin of the immune disturbance often lies deeper within the metabolic systems of the body.
Mitochondrial Dysfunction and Immune Activation
To understand why mitochondrial health matters in rheumatoid arthritis, it helps to examine how immune cells generate energy.
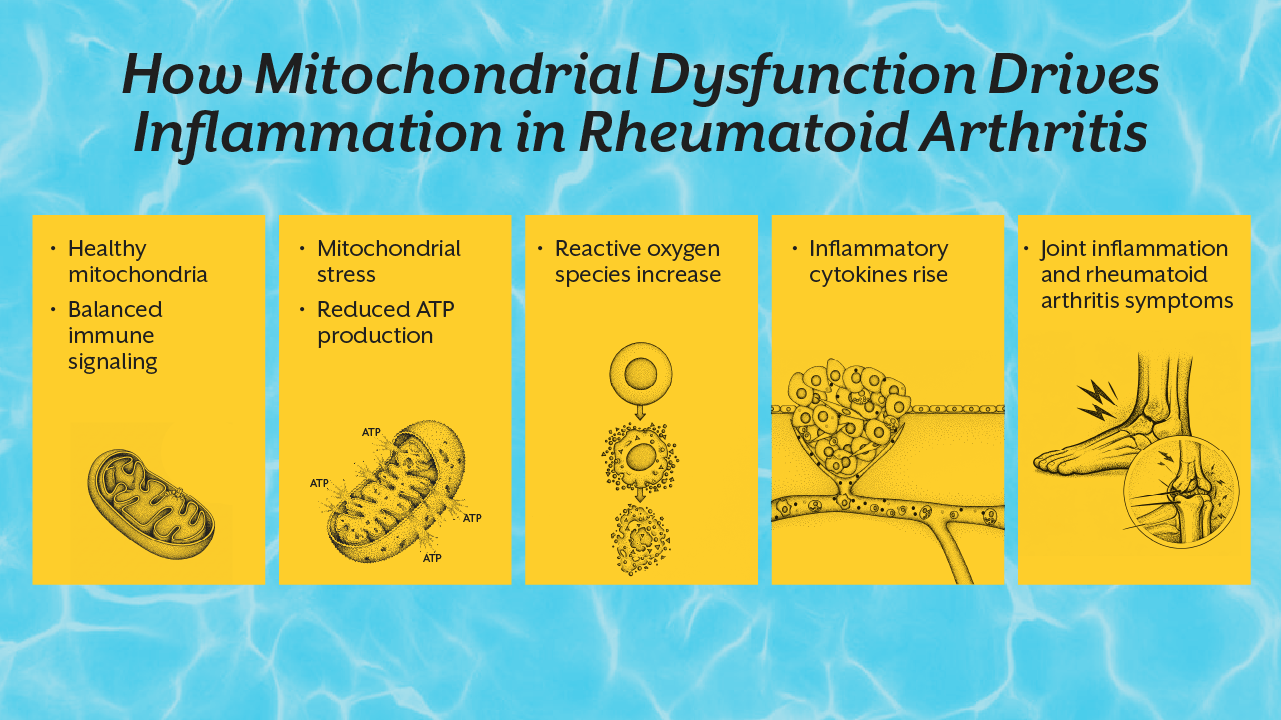
Immune cells rely on mitochondria to produce ATP which powers cellular activity. When mitochondria function normally, immune responses remain balanced and regulated.
However mitochondrial dysfunction can shift immune metabolism toward a state known as inflammatory metabolic reprogramming. In this state immune cells rely more heavily on glycolysis rather than efficient mitochondrial respiration. This metabolic change promotes excessive inflammatory signaling.
Several studies have identified mitochondrial abnormalities within synovial cells of individuals with rheumatoid arthritis. These cells show signs of oxidative stress, impaired mitochondrial respiration, and reduced energy production. The consequences extend beyond the joints.
Low cellular energy also contributes to fatigue, reduced physical resilience, and slow tissue repair. Many individuals with autoimmune arthritis report persistent exhaustion long before joint damage becomes severe.
This suggests that mitochondrial dysfunction may act as an early driver that shapes the inflammatory environment in rheumatoid arthritis.
The Overlooked Role of Metabolic Inflammation
Another hidden driver of inflammation in rheumatoid arthritis involves metabolic signals that influence immune activity.
Many individuals with autoimmune arthritis demonstrate metabolic abnormalities including impaired glucose regulation, oxidative stress, and altered lipid metabolism. These metabolic disturbances produce inflammatory signals that can worsen autoimmune reactions.
Inflammation in rheumatoid arthritis is therefore not only the result of immune malfunction. It is also shaped by the metabolic environment in which immune cells operate.
Mitochondrial stress plays a central role in this process. When mitochondria struggle to generate energy efficiently they release reactive oxygen species. These molecules activate inflammatory pathways that amplify immune activity. Over time this environment increases tissue vulnerability and accelerates joint inflammation.
This is one reason arthritis pain management strategies that focus only on pain suppression rarely address the full biological picture.

Why Fatigue Is Often the First Symptom
One of the most confusing aspects of rheumatoid arthritis symptoms is the profound fatigue experienced by many patients.
Fatigue in autoimmune arthritis is frequently dismissed as a side effect of chronic pain. However metabolic research suggests a different explanation.
When mitochondrial energy production declines, tissues throughout the body receive less cellular energy. Muscles, brain cells, and immune cells all depend on mitochondrial ATP production. Reduced ATP availability leads to widespread exhaustion.
This fatigue can appear years before significant joint damage develops. It may be accompanied by brain fog, poor exercise tolerance, or prolonged recovery after physical activity.
These early signs suggest that rheumatoid arthritis may be part of a broader systemic energy imbalance rather than a disease that begins solely in the joints.
A Systems Based Approach to RA Treatment
Understanding these hidden drivers opens the door to a broader view of RA treatment.
Traditional approaches often focus primarily on suppressing inflammation through medications. While these treatments can reduce symptoms, they may not address the metabolic conditions that influence immune behavior.
A systems approach examines the body as an interconnected network where immune function, mitochondrial energy production, and metabolic health influence one another. At iThrive Alive this perspective guides the evaluation process used during a root cause analysis. Rather than focusing only on the joints, the analysis explores deeper biological systems including metabolic function, nutrient status, inflammatory markers, and cellular energy metabolism.
This type of evaluation can help identify factors that may contribute to ongoing inflammation in rheumatoid arthritis. For individuals navigating autoimmune arthritis, understanding these underlying influences can provide valuable insight into their condition.
Lifestyle and Nutritional Influences on Cellular Energy
Mitochondrial function is highly sensitive to lifestyle patterns.
Sleep quality, dietary composition, physical activity, and environmental stressors all influence mitochondrial health. When these factors are optimized, mitochondrial energy production can improve and inflammatory signaling may decline. Many individuals with rheumatoid arthritis benefit from approaches that support cellular energy metabolism. These strategies may include anti inflammatory dietary patterns, nutrient repletion, and targeted supplementation that supports mitochondrial pathways.
Some supplements for rheumatoid arthritis are explored for their ability to influence oxidative stress and mitochondrial efficiency. While supplements alone cannot treat autoimmune arthritis, they may play a supportive role when combined with broader lifestyle strategies.
Within the iThrive Alive framework these interventions are implemented through the structured 3 months Alive program, which focuses on metabolic restoration through lifestyle interventions, smart supplementation, and dietary protocols. The goal is not simply arthritis pain management. The deeper objective is restoring the biological environment in which the immune system operates.

Key Takeaway
Rheumatoid arthritis is often described as a disease of the joints, but the biology behind the condition is far more complex. The immune system does not operate independently from metabolism or cellular energy systems. Emerging research suggests that mitochondrial dysfunction may play a significant role in shaping the inflammatory environment associated with autoimmune arthritis. When cellular energy systems begin to struggle, immune regulation changes and inflammatory signaling increases. Over time this environment can contribute to the development and progression of rheumatoid arthritis symptoms. By exploring the deeper metabolic drivers behind inflammation, it becomes possible to view RA treatment through a broader systems perspective that examines immune health, energy metabolism, and lifestyle factors together.


.svg)
.svg)















.webp)