



.svg)




.svg)
Modern weight management conversations often revolve around calories, discipline, and behavioral control. Yet clinical metabolic research increasingly reveals that appetite, fat storage, and energy expenditure are not merely behavioral outcomes, they are biologically regulated processes governed by cellular signaling networks.
Among the most powerful signaling molecules currently studied in metabolic medicine are therapeutic peptides. These short chains of amino acids function as highly specific biological messengers that instruct cells to modify metabolic activity, regulate appetite, influence energy utilization, and in some cases accelerate fat loss.
Unlike traditional metabolism boosters or appetite suppressants that act broadly across systems, therapeutic peptides operate through receptor-level communication. They do not simply stimulate or suppress, rather they inform. They alter how the body perceives energy sufficiency, nutrient availability, and metabolic demand.
This is precisely why peptides for weight loss are receiving increasing attention. However, at a clinical level, their role is far more nuanced than rapid fat reduction. They interact with insulin signaling, gut-brain communication, inflammatory pathways, and neuroendocrine regulation.
At iThrive, peptide therapy is never viewed as an isolated intervention. It is examined within the broader landscape of metabolic health, cellular repair, and root-cause physiology, the same systems discussed in our published articles “Hyperinsulinemia: Hidden Cause of Weight Gain and Diabetes” and “How I lost 16 kgs in 4 months - Mugdha Pradhan’s easy weight loss journey with GLP agonists.”
To understand why therapeutic peptides are powerful and why they must be used responsibly, we must first understand how they communicate within the body.
The human body functions through layered communication systems. Hormones transmit broad systemic messages. Neurotransmitters coordinate rapid responses. Peptides, however, deliver precision instructions.
They bind to specific cellular receptors, activating intracellular signaling cascades that modify gene expression, enzyme activity, and energy utilization. This receptor specificity is what allows peptides for fat loss to influence appetite regulation without uniformly stimulating the entire endocrine system.
When metabolic imbalance develops , particularly insulin resistance, chronic inflammation, or stress-driven hormonal disruption and communication between organs becomes inefficient. The brain misinterprets energy availability. Fat tissue releases altered signaling molecules. The liver increases glucose production. Hunger signals intensify despite adequate energy stores.
Several therapeutic peptides that influence appetite regulation and metabolic signaling are being widely researched and discussed in metabolic medicine. These include:
• Semaglutide - a GLP-1 receptor agonist commonly studied for appetite regulation and glucose control.
• Tirzepatide (Mounjaro) - a dual GIP and GLP-1 receptor agonist that influences insulin signaling and satiety pathways.
• Retatrutide - a multi-pathway peptide that interacts with GLP-1, GIP, and glucagon signaling systems.
• BPC-157 - often discussed in regenerative medicine research related to tissue repair and inflammation modulation.
• TB-500 (Thymosin Beta-4) - studied in recovery and tissue regeneration contexts.
These molecules are being investigated for their ability to influence appetite signaling, metabolic efficiency, tissue repair, and energy regulation. Their physiological effects depend heavily on individual metabolic context, dosage, and clinical supervision. They should always be understood as regulatory signaling tools rather than standalone metabolic solutions.
Therapeutic peptides act as corrective signals within this disrupted communication network. Rather than forcing metabolic change, they help recalibrate signaling pathways that regulate satiety, nutrient partitioning, and fuel utilization.
Appetite is not a psychological phenomenon alone, it is rather a neuroendocrine process governed by the gut-brain axis. Signals from intestinal cells, adipose tissue, and circulating nutrients are integrated in hypothalamic centers that determine hunger intensity and satiety duration.
Certain weight loss peptides interact directly with this regulatory axis. They influence gastric emptying, satiety hormone release, and reward-driven food behavior. The result is not forced restriction but reduced physiological drive to eat.
Clinically, this effect is often described as reduction in food noise, the persistent cognitive and sensory pull toward food that many individuals with metabolic dysregulation experience continuously.
However, appetite suppression alone is not metabolic healing. Without restoration of insulin sensitivity, mitochondrial function, and inflammatory balance, appetite regulation remains temporary. This distinction is central to the iThrive Alive approach.
Among newer metabolic peptides, multi-pathway signaling molecules are receiving increasing scientific interest because they influence more than one regulatory axis simultaneously. Rather than acting only on hunger signals, some peptides also affect insulin response and energy expenditure demonstrating how interconnected metabolic regulation truly is.
Beyond appetite, therapeutic peptides influence how the body processes and uses energy.
Metabolism is not simply calorie burning. It is the orchestration of glucose handling, fat oxidation, insulin signaling, and mitochondrial activity. In metabolic dysfunction, energy is stored inefficiently, oxidized poorly, and often diverted into fat accumulation.
Certain peptides for weight management solutions modify nutrient partitioning, the biological decision of whether incoming energy is stored or utilized. They may improve insulin sensitivity, enhance lipolysis, or increase cellular responsiveness to metabolic demand.
This is why some individuals experience improved body composition even when calorie intake changes modestly. The metabolic environment itself becomes more efficient.
Metabolism does not operate independently from structural health. Muscle tissue, connective tissue, and organ integrity all influence energy regulation.
Peptides used in regenerative medicine such as those supporting ligament, tendon, and muscle healing indirectly influence metabolic function by improving movement capacity, reducing chronic inflammation, and restoring physical activity tolerance.
Repair-oriented peptides signal angiogenesis, collagen synthesis, and tissue remodeling. When injury recovery accelerates, physical activity becomes sustainable, which further improves metabolic health.
This illustrates an important principle: fat loss peptides do not act in isolation. Structural repair, inflammation control, and metabolic regulation are interdependent processes.
Mugdha Pradhan, founder of iThrive experienced significant weight gain despite advanced nutritional knowledge. Emotional stress, nervous system overload, and persistent food-seeking behavior created metabolic conditions resistant to lifestyle change alone.
After extensive research into peptide physiology and metabolic signaling, targeted peptide therapy was introduced alongside structured nutrition, resistance training, micronutrient optimization, and nervous system regulation.
Over the years, Mugdha’s own health journey has evolved alongside her clinical work. Like many practitioners deeply invested in longevity and metabolic health, she has explored advanced regenerative tools including peptides in a structured, medically supervised manner.
Her transformation, however, was not about dramatic overnight change. It was about improving recovery, maintaining lean muscle, optimising metabolic markers, and supporting cellular repair. Peptides, when used appropriately and under guidance, can support signalling pathways related to regeneration, inflammation control, and tissue healing. But they are never a shortcut; they are adjuncts to disciplined nutrition, sleep, strength training, and stress regulation.
For Mugdha, the real shift wasn’t aesthetic; it was physiological. Better recovery, improved energy stability, sharper focus, and sustained muscle quality were markers that her internal systems were functioning more efficiently. The visible changes were a by-product of internal repair.
The outcome was not merely weight reduction but improved insulin sensitivity, decreased visceral fat, and restoration of appetite regulation. The intervention worked not because peptides replaced lifestyle but because they enabled adherence to metabolic healing protocols.
She is also clear that peptides are not for everyone and not a replacement for foundational health work. Gut health, micronutrient sufficiency, hormonal balance, and lifestyle remain the core pillars. Advanced tools only work when the basics are in place.
In Mugdha’s case, the therapeutic peptide used was Retatrutide, selected after extensive review of metabolic pathway research and clinical data.
Three biological factors made Retatrutide particularly relevant for her metabolic condition:
GLP-1 Pathway - Appetite and Satiety Regulation
Supported reduction in food noise, improved fullness signaling, and enhanced dietary adherence.
GIP Pathway - Insulin Efficiency and Nutrient Utilization
Helped improve glucose handling, metabolic flexibility, and hormonal coordination of energy storage.
Glucagon Pathway - Energy Expenditure and Fat Mobilisation
Supported increased metabolic activity and utilization of stored energy, contributing to body-composition change rather than intake reduction alone.
This multi-pathway signaling profile is what distinguished Retatrutide from earlier single-pathway metabolic peptides in her research process.
In her view, longevity is not about chasing biohacks, it’s about supporting the body’s natural regenerative intelligence with responsibility and context.
Her detailed experience is documented in the video:
“How I lost 16 kgs in 4 months - Mugdha Pradhan’s easy weight loss journey with GLP agonists.”
MOST IMPORTANT: It is important to note that the therapeutic effect observed was not attributed to the peptide alone. The intervention worked within a structured physiological framework that included nutrition planning, resistance training, micronutrient sufficiency, and nervous system regulation. The peptide functioned as a signaling facilitator that improved adherence and metabolic responsiveness.

Not all cellular signaling molecules operate similarly.
Therapeutic peptides typically accelerate biological processes. They stimulate pathways involved in repair, metabolism, or appetite control.
Bioregulators, in contrast, influence gene expression patterns more subtly. They restore cellular memory guiding tissues toward long-term functional balance rather than rapid activation.
In clinical decision-making, peptides are often used when rapid metabolic intervention is required, while bioregulators support sustained cellular normalization.
Understanding this distinction prevents misuse of powerful metabolic tools.

Despite their powerful signaling effects, peptides for fat loss cannot compensate for poor nutritional status or metabolic dysfunction.
Protein deficiency, micronutrient insufficiency, circadian disruption, chronic stress, and inflammatory load all determine whether peptide signals translate into meaningful physiological change.
This is why iThrive Alive emphasizes foundational metabolic restoration such as nutrition planning, lifestyle restructuring, and targeted supplementation before advanced interventions.
Peptides amplify biological potential. They do not create it.
At iThrive Alive, peptide therapy is never approached as a universal weight management solution.
Clinical assessment evaluates fasting insulin, inflammatory markers, lipid patterns, stress physiology, sleep architecture, and nutrient status. Only when underlying metabolic drivers are identified does targeted intervention become meaningful.
This root-cause methodology ensures peptides are used as strategic metabolic tools, and not shortcuts.
Therapeutic peptides represent one of the most advanced developments in metabolic medicine because they function at the level of cellular communication. They influence appetite regulation, energy utilization, tissue repair, and neuroendocrine signaling with remarkable precision. Yet their true value lies not in rapid fat loss but in restoring biological responsiveness within systems already supported by nutrition, lifestyle, and metabolic correction. When used responsibly and within a root-cause framework, peptides can help recalibrate dysfunctional signaling networks that drive persistent weight gain. When used without metabolic foundation, they remain temporary metabolic accelerators rather than long-term solutions.
For clinical screening, metabolic assessment, or guidance on whether advanced peptide-based interventions are appropriate for your physiology, you may contact our team directly.
Email: care@ithrive.shop
📲 WhatsApp us directly for metabolic screening
All interventions are recommended only after structured metabolic evaluation and suitability screening.




.svg)
Most conversations around obesity reduce overeating to a psychological weakness. The narrative is simple: stress leads to emotional eating, emotional eating leads to excess calories, and excess calories lead to weight gain. While emotions absolutely influence food behavior, this explanation is incomplete and often misleading.
At iThrive Alive, we frequently see individuals who insist they are “not overeating much,” yet weight continues to increase. Others describe relentless hunger, evening cravings, or an inability to feel satisfied even after a full meal. The assumption is often emotional fragility. The reality, more often than not, is metabolic imbalance.
Overeating in obesity is rarely just about comfort food. It is frequently the downstream effect of insulin resistance and obesity interacting with leptin resistance, disrupted satiety signaling, circadian misalignment, and cortisol-driven energy shifts. What appears as lack of discipline is often hormonal imbalance and weight gain unfolding in real time.
Understanding this distinction changes everything. Because if overeating is a signal of metabolic chaos rather than character weakness, the solution cannot be shame. It must be physiology.
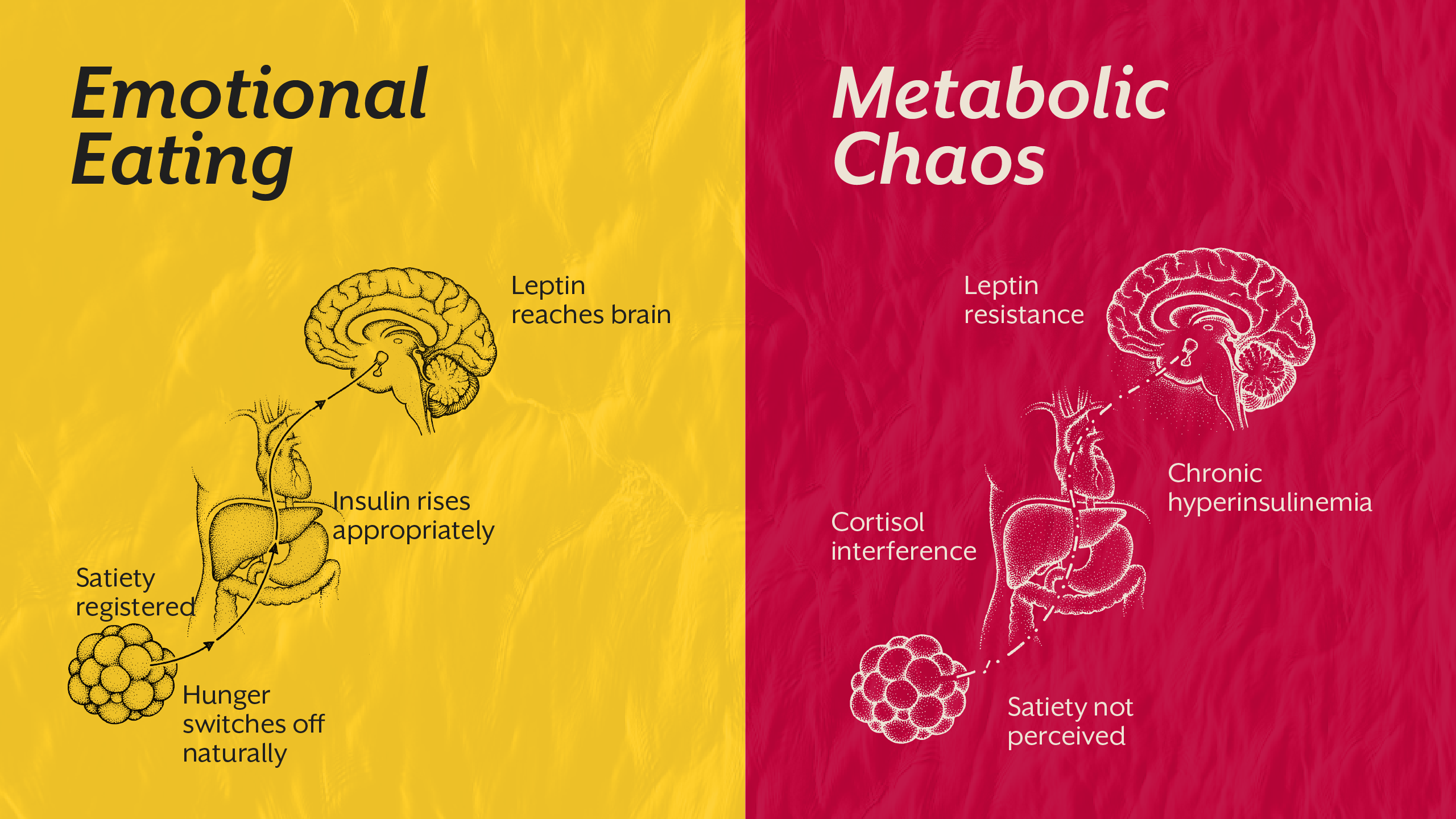
Hunger is regulated through an intricate network involving the gut, adipose tissue, pancreas, liver, and brain. Hormones such as insulin, leptin, ghrelin, and cortisol constantly communicate to determine whether we seek food or feel satisfied.
In a metabolically healthy individual, this system works with remarkable precision. After eating, insulin rises appropriately, glucose is stored or utilized, leptin signals energy sufficiency to the hypothalamus, and hunger naturally declines. Energy intake and expenditure remain balanced without conscious effort.
In metabolic imbalance, however, this regulatory loop begins to fail. Insulin resistance disrupts cellular energy access. Leptin resistance impairs the brain’s ability to sense stored fat. Cortisol alters glucose production and appetite patterns. The result is a body that behaves as if it is deprived, even when energy stores are abundant.
This is where overeating begins to shift from psychological narrative to biological survival response.

One of the most overlooked drivers of overeating in obesity is insulin resistance. When cells become less responsive to insulin, glucose cannot efficiently enter muscle and liver tissue. Despite adequate energy intake, cells behave as though they are under-fueled.
This creates what we call an “energy lock.” Fat cells remain in storage mode, unable to release fuel effectively. The brain interprets this restricted fuel access as scarcity, triggering increased appetite.
In this context, overeating is not indulgence. It is a compensatory response to cellular energy dysfunction. This explains why individuals with insulin resistance and obesity often experience strong carbohydrate cravings or frequent hunger shortly after meals.
We explored the early compensatory phase of hyperinsulinemia in our blog, “Fasting Insulin vs Fasting Glucose: Which One Actually Predicts Type 2 Diabetes?” That silent elevation in insulin often precedes overt metabolic disease, and it also precedes relentless appetite dysregulation.
Leptin is produced by fat tissue and signals satiety to the brain. In theory, higher body fat should mean stronger leptin signaling and reduced appetite. In obesity, the opposite frequently occurs.
Leptin resistance develops when the hypothalamus no longer responds adequately to leptin’s message. Despite high circulating leptin levels, the brain perceives energy insufficiency. Hunger remains elevated, and energy expenditure may decline.
This creates a paradox: more stored fat, yet persistent drive to eat.
In our earlier exploration of hyperinsulinemia as a hidden driver of metabolic dysfunction, we emphasized how hormonal feedback loops become distorted long before overt disease. Leptin resistance fits squarely within this broader metabolic imbalance narrative.
Overeating, in this case, is a brain-level communication failure, not emotional fragility.
While insulin and leptin dominate the conversation, cortisol plays a crucial amplifying role. Chronic stress elevates cortisol, which increases hepatic glucose output and can promote insulin resistance over time.
Elevated cortisol also alters appetite regulation, often increasing cravings for energy-dense foods. Evening overeating patterns frequently correlate with disrupted circadian cortisol rhythms rather than emotional instability alone.
In this scenario, what appears to be “stress eating” is partially neuroendocrine recalibration gone wrong. Cortisol shifts fuel allocation, impairs sleep, and disrupts appetite hormones. The body attempts to restore stability through increased intake.
Understanding cortisol and weight gain reframes emotional eating as, in many cases, stress-driven metabolic adaptation.
Emotional eating certainly exists. However, not all overeating stems from emotional distress. Distinguishing between psychological triggers and physiological dysregulation is critical.
True emotional eating tends to be situational, episodic, and tied to specific emotional states. Metabolic-driven overeating is often persistent, characterized by strong hunger signals, reduced satiety, and physical symptoms such as energy crashes or sugar cravings.
At iThrive Alive, we rarely stop at surface narratives. We assess fasting insulin, inflammatory markers, lipid patterns, sleep rhythms, and stress physiology. Root cause analysis reveals whether overeating is primarily behavioral or hormonally driven.
Without this differentiation, interventions fail. Dieting strategies aimed at behavior alone cannot correct hormonal imbalance and weight gain driven by metabolic dysfunction.
Emerging obesity science highlights the role of low-grade inflammation in appetite dysregulation. Inflammatory cytokines can impair insulin signaling and disrupt hypothalamic appetite control.
Mitochondrial dysfunction further complicates energy sensing. When cellular energy production declines, the body increases hunger signals to compensate. This explains why some individuals overeat despite consuming calorie-dense diets, they are rather energy-replete but metabolically inefficient.
This deeper understanding of obesity and hormones challenges simplistic calorie-based explanations. Obesity is not merely about intake. It is about energy sensing and allocation gone awry.
If overeating is driven by metabolic chaos, the solution must target underlying physiology. At iThrive, interventions focus on restoring insulin sensitivity, improving leptin signaling, recalibrating cortisol rhythms, and enhancing mitochondrial efficiency.
This involves structured meal sequencing, protein prioritization, micronutrient optimization, circadian alignment, and targeted supplementation. Smart eating is not restriction; it is strategic metabolic recalibration.
We also evaluate stress physiology, sleep architecture, and inflammatory load. Because addressing insulin resistance and obesity without correcting cortisol patterns or inflammatory triggers leads to incomplete recovery.
For individuals unsure whether their overeating is emotional or metabolic, a clinical review can clarify the drivers and guide precision intervention.
Perhaps the most important shift is conceptual. Obesity is not simply excess weight. It is a survival response to chronic metabolic stress.
When insulin resistance, leptin resistance, and cortisol dysregulation converge, the body prioritizes storage and hunger. Overeating becomes an adaptive attempt to restore perceived energy deficits.
Understanding this changes the emotional burden carried by individuals struggling with weight. The problem is not weakness. It is metabolic signaling gone wrong.
At iThrive Alive, this understanding shapes everything. We move beyond obesity as a behavior problem and treat it as a systems-level disorder requiring structured, physiology-first intervention.
Overeating in obesity is not always emotional fragility; it is frequently the outward expression of internal metabolic chaos. Insulin resistance locks energy inside cells, leptin resistance prevents the brain from recognizing stored fat, and cortisol reshapes appetite under chronic stress. Together, these hormonal disruptions create persistent hunger, cravings, and fat storage patterns that feel uncontrollable. When we shift the lens from willpower to physiology, solutions become clearer and more compassionate. True progress in obesity requires restoring metabolic balance, not merely suppressing appetite. At iThrive Alive, this root-cause perspective transforms overeating from a moral narrative into a solvable biological problem.




.svg)
For years, obesity has been framed as a consequence of excess eating and insufficient movement. Yet in clinical practice, this explanation repeatedly collapses. Individuals follow structured diets, restrict calories, exercise diligently, and still experience progressive weight gain, fatigue, intense hunger, and metabolic slowdown. These patterns reveal a deeper truth: obesity is rarely a behavioural issue first. It is a hormonal one.
Two hormones dominate this conversation that are insulin and leptin. Both are central regulators of energy storage, appetite, and metabolic flexibility. When either hormone stops communicating effectively with the body, fat loss resistance emerges. But a critical clinical question remains under-explored: which resistance comes first, insulin resistance or leptin resistance?
Understanding the sequence matters because it determines how obesity develops, why diabetes risk escalates, and why generic diet plans fail. At iThrive Alive, obesity and diabetes are approached as adaptive metabolic states driven by disrupted hormonal signalling, not as isolated diseases. This article examines insulin resistance vs leptin resistance through a systems biology lens, explaining how they interact, which tends to appear first, and how root-cause interventions restore metabolic intelligence.
Insulin is a storage and signalling hormone released by the pancreas in response to rising blood glucose. Its primary job is to escort glucose into muscle, liver, and fat cells, where it can be used or stored for later energy. Under healthy conditions, insulin rises briefly after meals and falls during fasting, allowing metabolic flexibility between glucose and fat usage.
Insulin resistance develops when cells become less responsive to insulin’s signal. Glucose remains trapped in the bloodstream, prompting the pancreas to secrete even more insulin. Over time, this compensatory hyperinsulinemia becomes chronic, creating a metabolic environment biased toward fat storage and inflammation.
This is why insulin resistance and obesity are so tightly linked. Elevated insulin does not merely reflect weight gain, it actively promotes it by inhibiting fat breakdown and encouraging adipose expansion.
A critical misconception is that insulin resistance equals diabetes. In reality, insulin resistance can exist silently for years before fasting glucose or HbA1c rise. During this phase, the body is still compensating, but at a cost: worsening fatigue, abdominal fat gain, post-meal crashes, and increased hunger.
From a clinical perspective, insulin resistance is often the earliest metabolic disturbance in hormonal obesity. It alters fuel partitioning long before appetite hormones derail conscious eating behaviour.

Leptin is secreted by fat cells and communicates with the hypothalamus to signal energy sufficiency. In a healthy system, rising fat stores increase leptin levels, reducing appetite and increasing energy expenditure. Leptin resistance occurs when the brain no longer responds appropriately to leptin’s signal, despite high circulating levels.
This is why individuals with obesity often have elevated leptin yet experience persistent hunger. Leptin resistance and obesity form a vicious cycle: fat gain increases leptin, but impaired signalling prevents appetite regulation.
Contrary to popular narratives, leptin resistance is rarely the initiating defect. It typically emerges after prolonged metabolic stress such as chronic insulin elevation, inflammation, disrupted circadian rhythms, and poor sleep. Insulin directly influences leptin transport across the blood-brain barrier, meaning insulin resistance can impair leptin signalling upstream.
This sequence explains why appetite dysregulation often appears after years of weight gain, not before.
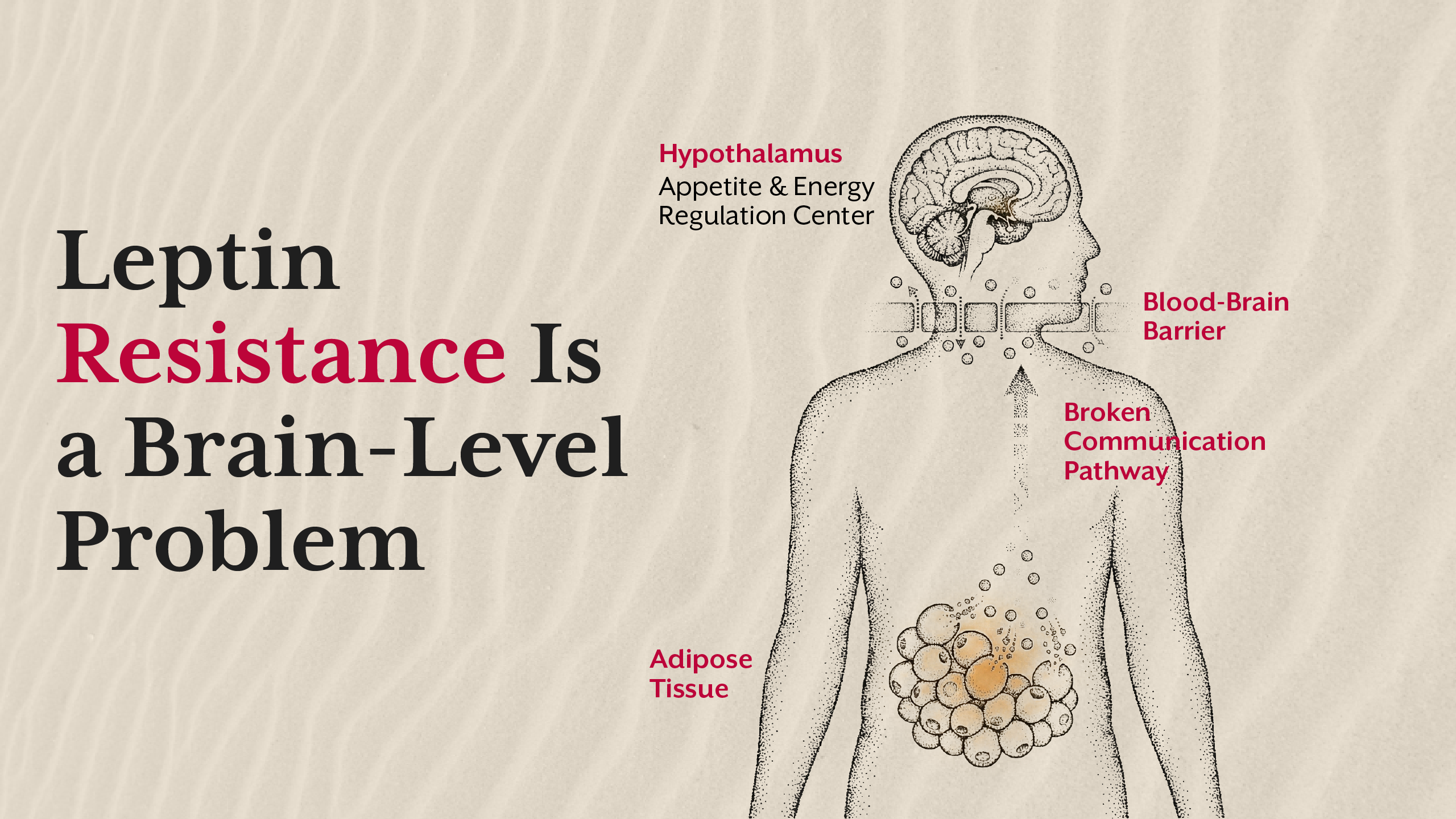
In clinical observation, insulin resistance typically precedes leptin resistance. Elevated insulin alters fat storage patterns, promotes inflammation, and disrupts central appetite signalling over time. Leptin resistance then amplifies weight gain by removing the brain’s braking system.
This distinction matters because treating leptin resistance without addressing insulin resistance often fails. Appetite control strategies collapse when metabolic signalling remains impaired.
Fat loss resistance occurs when the body actively defends stored fat due to perceived metabolic threat. Chronic insulin elevation blocks lipolysis, while leptin resistance removes satiety cues. Together, they create a state where calorie restriction increases stress hormones, slows metabolism, and worsens outcomes.
This is why obesity and hormones must be addressed together. Without restoring signalling pathways, weight loss attempts reinforce the very adaptations driving weight gain.
At iThrive Alive, obesity management focuses on reversing the drivers of insulin and leptin resistance through structured lifestyle interventions, targeted supplementation, and personalized eating strategies. Rather than suppressing symptoms, clinical reviews identify metabolic bottlenecks, circadian disruption, nutrient deficiencies, and inflammatory triggers.
Root-cause assessment allows insulin signalling to normalise first, creating the conditions for leptin sensitivity to recover naturally. Appetite stabilisation, energy improvement, and sustainable fat loss follow as outcomes, not goals forced through restriction.
Insulin resistance vs leptin resistance is not a debate of equals. Insulin resistance usually initiates the metabolic cascade that leads to leptin resistance, fat loss resistance, and obesity progression. Treating obesity without addressing this sequence leads to frustration and relapse. When hormonal communication is restored, weight regulation becomes a biological response rather than a daily struggle.




.svg)
Most people believe obesity begins when weight visibly increases. Clinically, it is often “diagnosed” when BMI crosses a cutoff or when clothes no longer fit the same way. But by the time this happens, the biological processes driving obesity have usually been active for years, and sometimes even decades.
At iThrive Alive, we do not view obesity as a sudden accumulation of fat. We see it as the final visible outcome of long-standing metabolic strain, hormonal compensation, and inflammatory signalling. Long before fat mass increases, the blood quietly records the body’s survival decisions.
These early changes don’t show up in routine check-ups focused only on fasting glucose or cholesterol totals. They appear in specific blood markers that reflect how the body is adapting to stress, energy excess, inflammation, and disrupted signalling. When interpreted correctly, these biomarkers can predict obesity risk well before weight gain begins.
This blog explains which blood markers predict obesity, why they change early, and how Alive uses them to intervene upstream, before the scale moves.
Obesity is often framed as a failure of willpower. Biologically, it is anything but. The human body prioritises survival over aesthetics. When faced with repeated metabolic stress such as overnutrition, poor sleep, inflammation, emotional strain, toxin exposure, it eventually adapts.
Fat tissue expands not as excess, but as insurance:
This adaptive phase is invisible on the weighing scale. What is visible, however, is the blood. Hormones rise, inflammatory signals increase, lipid handling shifts, and insulin demand climbs. These changes precede weight gain and form the true early signs of obesity in blood tests.
This reframing is central to how Alive approaches obesity science: weight gain is not the cause, it is rather the consequence.
Fasting insulin is one of the most underutilised yet powerful biomarkers of obesity risk. While glucose reflects the result of regulation, insulin reflects the effort required to maintain that regulation.
When cells begin responding poorly to insulin, the pancreas compensates by producing more. Blood glucose may remain normal, but insulin rises quietly. This state, that is hyperinsulinemia, is not benign. It actively promotes fat storage and suppresses fat release.
Elevated fasting insulin years before weight gain signals:
Alive’s earlier blog on hyperinsulinemia as a hidden driver of metabolic disease explores how this state often exists undetected in “healthy” individuals. In obesity prediction, fasting insulin is foundational.
Low-grade, chronic inflammation is a consistent predictor of future obesity. Markers such as high-sensitivity CRP (hs-CRP) often rise well before fat mass increases.
Inflammation alters insulin signalling, disrupts appetite regulation, and encourages fat storage as a protective buffer. Importantly, this is not acute illness inflammation, rather it is metabolic inflammation driven by:
At Alive, inflammation is never viewed in isolation. We map it alongside insulin, lipid patterns, and lifestyle context to understand why the body has entered a defensive state.
Standard lipid panels often miss early obesity risk because totals can look “normal.” The real signal lies in patterns, especially:
A rising triglyceride-to-HDL ratio reflects impaired fat oxidation and insulin resistance. It signals that the body is struggling to clear circulating energy efficiently and is redirecting it toward storage.
This lipid dysregulation is an early obesity blood marker, not merely a cardiovascular one.

Mild elevations in ALT and AST, even within “normal” ranges, often reflect early fat accumulation in the liver. Non-alcoholic fatty liver disease is not a consequence of obesity, it is frequently a precursor.
The liver governs glucose output, fat metabolism, and insulin clearance. When liver cells accumulate fat, insulin resistance accelerates system-wide.
Alive often sees liver markers shift long before individuals consider themselves overweight.
Leptin is often called the “satiety hormone,” but its dysfunction is deeply misunderstood. Rising leptin levels indicate that the brain is no longer responding appropriately to fullness signals.
Leptin resistance develops early in metabolic dysfunction and contributes to:
High leptin is not a lack of discipline, it is rather a communication failure between fat tissue and the brain.
No single blood test predicts obesity in isolation. The power lies in patterns across systems:
This pattern-based interpretation is core to Alive’s root-cause analysis approach. Rather than asking “Is this value abnormal?”, we ask “What survival adaptation is this value reflecting?”

Identifying obesity risk blood markers is not about labelling disease—it is about timing intervention correctly. When addressed early, metabolic adaptations are reversible.
Alive focuses on:
This is achieved through lifestyle alignment, smart supplementation, and signal-based eating, not calorie obsession. For individuals unsure which marker is driving their pattern, a structured root-cause clinical review helps identify the dominant lever.
Obesity does not appear suddenly, and it does not begin with weight gain. It begins as a series of quiet biochemical adaptations visible in blood long before the mirror reflects change. Fasting insulin, inflammatory markers, lipid ratios, liver enzymes, and hormonal signals together tell a story of a body trying to survive modern stressors. When these markers are interpreted early and in context, obesity becomes predictable, and more importantly, preventable. At iThrive Alive, this shift from reacting to weight gain toward understanding metabolic signals is what allows meaningful, lasting reversal rather than temporary weight loss.




.svg)
For decades, obesity has been framed as a personal failure like too much food, too little discipline, not enough movement. This narrative has shaped public health messaging, diet culture, and even clinical conversations. Yet despite an explosion of calorie-tracking apps, fitness programs, and “fat-loss hacks,” obesity rates continue to rise globally. If willpower were the real issue, this problem should have been solved by now.
At iThrive Alive, we approach obesity differently. We don’t see excess weight as a moral flaw or a lack of effort. We see it as a survival response, rather as a biological adaptation to internal chaos. When the body perceives threat, instability, or energy insecurity, it does what it has always done throughout human history: it stores.
To understand obesity properly, we must move away from blame and toward biology. This blog explores obesity science through the lens of metabolism, insulin signaling, inflammation, and evolutionary survival. More importantly, it explains why obesity is not about willpower, and why lasting change only occurs when root causes are addressed.
Weight loss focuses on the scale. Obesity science focuses on why the scale changed in the first place. Obesity is not simply excess fat mass; it is a state of metabolic dysfunction where the body has lost flexibility in how it uses and stores energy.
From a biological standpoint, obesity reflects:
This is why two people can eat similar diets and live similar lifestyles yet experience drastically different weight outcomes. Obesity causes are not uniform; they are deeply individual and metabolically driven.
When obesity is reduced to “eat less, move more,” we ignore the fact that metabolism adapts. Calorie restriction without metabolic repair often worsens the problem by increasing stress hormones, slowing energy expenditure, and reinforcing fat storage.
The human body evolved to survive scarcity, infection, and environmental stress. When modern stressors like processed food, sleep disruption, chronic inflammation, toxins, emotional strain mimic these threats, the body responds in the same way it always has: by conserving energy.
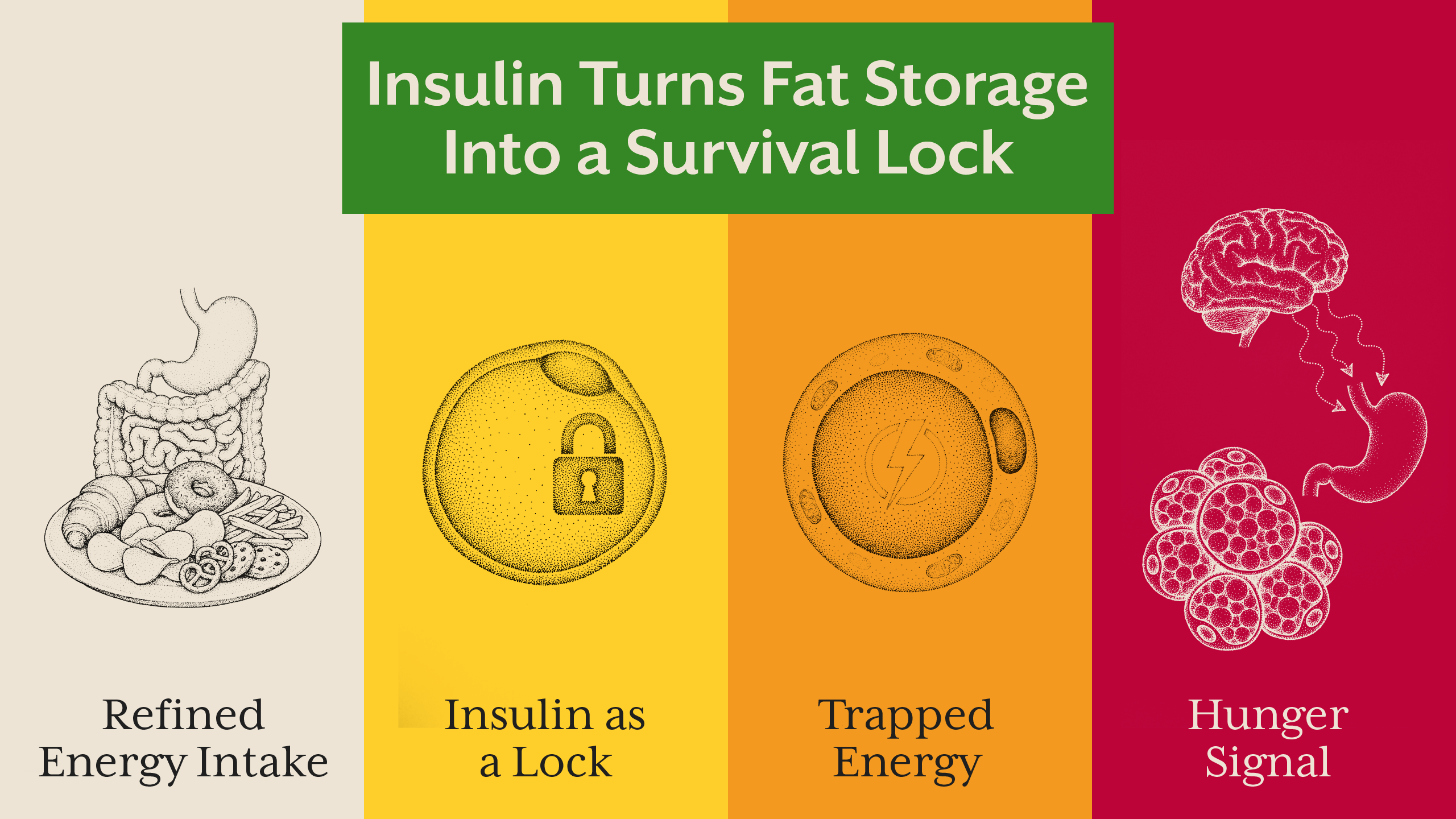
Insulin plays a central role here. Chronically elevated insulin acts as a metabolic lock, preventing fat breakdown and signaling the body to store incoming energy. This state is often present years before diabetes is diagnosed, a concept explored in our blog “Hyperinsulinemia: The Hidden Driver of Metabolic Disease” on Alive.
Importantly, obesity is often a protective adaptation, not a malfunction. Fat tissue acts as:
From a survival perspective, this makes sense. From a modern health perspective, it becomes problematic.
Willpower assumes conscious control. Obesity science shows that most eating behavior is hormonally driven. Insulin, leptin, ghrelin, cortisol, and dopamine shape hunger, cravings, and satiety long before conscious thought enters the picture.
When insulin remains high:
This is why individuals with obesity are often biologically hungrier, not weaker. Blaming willpower ignores the neurological reality of metabolic disease.
At iThrive Alive, we frequently see individuals who have tried “everything” such as clean eating, intermittent fasting, intense workouts yet remain stuck. When we correct insulin signaling, gut health, micronutrient deficiencies, and circadian rhythm, hunger normalizes without force.
This reframing is central to obesity myths versus obesity science.
Obesity causes rarely exist in isolation. At Alive, root cause analysis often reveals overlapping contributors:
This is why obesity often coexists with PCOS, fatty liver, insulin resistance, and thyroid dysfunction. Treating weight alone misses the broader metabolic picture.
Our approach mirrors what we discussed in “Insulin Resistance: The Silent Phase Before Disease” long before lab values cross diagnostic thresholds, the body is compensating.
At iThrive Alive, obesity reversal is not about dieting, it’s about changing the signal. Through lifestyle interventions, smart supplementation, and precise food sequencing, we restore metabolic flexibility.
Key principles include:
This approach is deeply individual. That’s why root cause assessment remains foundational. For those seeking structured guidance, a clinical review helps identify which systems are driving weight retention.

Obesity is not a personal failure, rather is a biological response to sustained metabolic stress. When the body perceives instability, it stores energy as protection. Lasting change occurs only when this perceived threat is removed. By addressing insulin signaling, inflammation, gut health, sleep, and nutrient status, the body no longer needs excess weight for survival. True obesity reversal is not forced; it is allowed.




.svg)
For decades, diabetes screening has revolved around a single, seemingly decisive number that is blood glucose. If fasting glucose falls within a laboratory-defined “normal” range, most individuals are reassured that their metabolic health is intact. This reassurance is often reinforced by annual health checkups, insurance screenings, and even public health messaging that equates diabetes risk almost entirely with sugar levels. Yet clinical reality tells a more uncomfortable story. A large proportion of people who eventually develop type 2 diabetes spend years, sometimes decades, being told that their glucose is normal.
This gap between reassurance and reality exists because glucose is not an early warning signal. It is a late-stage outcome. The metabolic system is remarkably adaptive and does not fail abruptly. Long before glucose begins to rise, the body compensates silently by producing more insulin to maintain glucose balance. This compensatory phase, marked by rising fasting insulin and progressive insulin resistance is where type 2 diabetes truly begins, even though it remains invisible on routine blood reports.
At iThrive, we do not view diabetes as a disease of sugar excess. We see it as a progressive failure of metabolic regulation. When viewed through this lens, insulin dynamics offer far greater insight into risk and trajectory than glucose values alone. Understanding the difference between fasting insulin and fasting glucose is therefore not a technical nuance, it is foundational to predicting, preventing, and reversing metabolic disease.
Fasting glucose measures the concentration of glucose circulating in the blood after an overnight fast. Clinically, it is used to identify overt disturbances in glucose regulation, commonly classified into normal, prediabetes, and diabetes ranges. On paper, this seems logical: if glucose is high, regulation must be impaired. However, this interpretation overlooks a critical question as to how much metabolic effort was required to keep glucose within range?
Fasting glucose reflects the end result of multiple physiological processes working together, including insulin secretion, hepatic glucose production, muscle uptake of glucose, and hormonal counter-regulation. What it does not capture is the strain placed on these systems to maintain balance. A normal glucose value can therefore exist in two very different metabolic realities: one where regulation is effortless, and another where regulation is maintained only through excessive insulin output.
The limitation is not the test itself, rather it’s what the test represents. Fasting glucose reflects the end result of multiple metabolic processes, not the strain those processes are under.

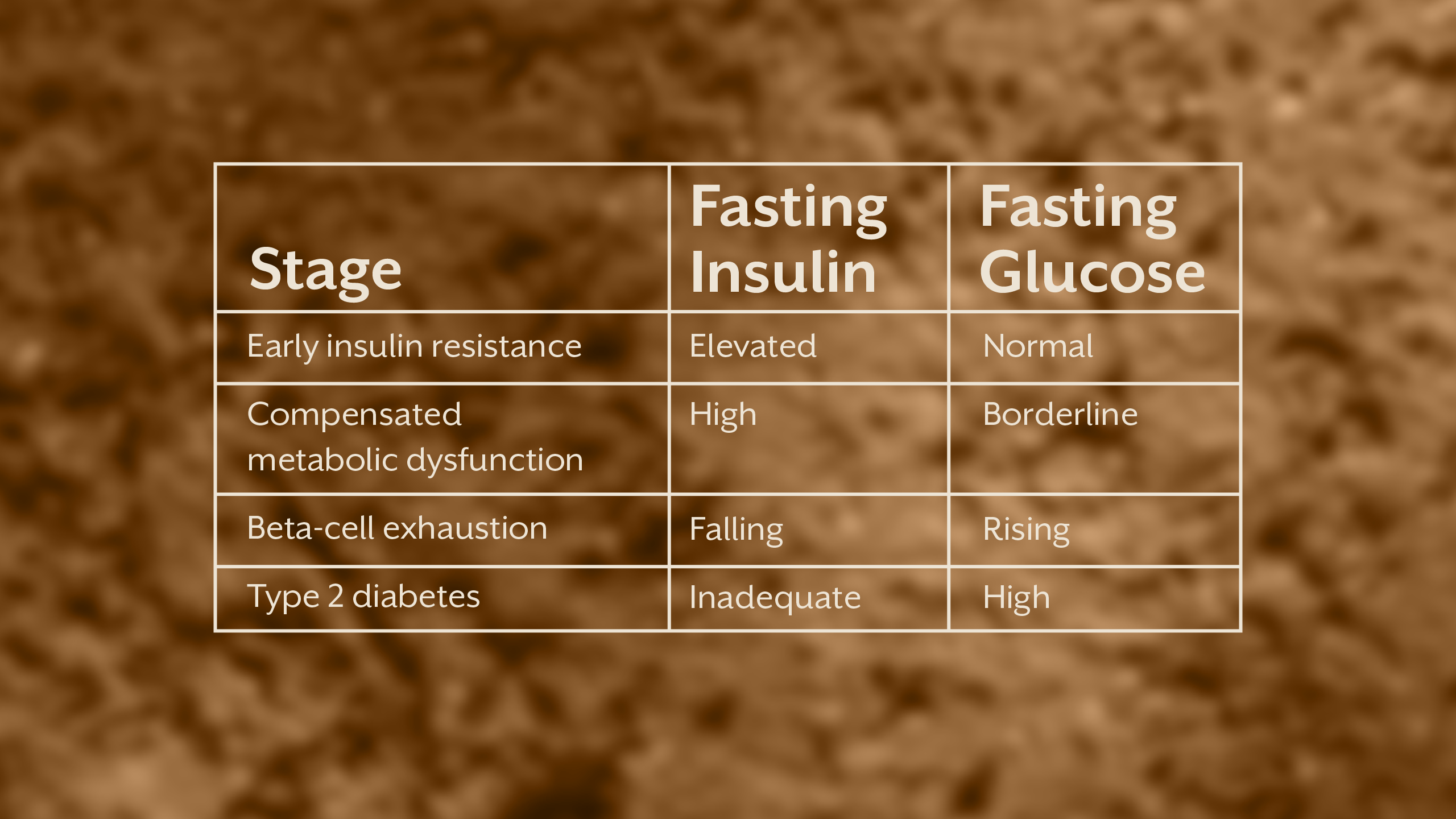
In the early stages of insulin resistance, cells particularly in muscle and liver become quite less responsive to insulin’s signal. To compensate, the pancreas increases insulin secretion. As long as pancreatic beta cells are capable of sustaining this higher output, glucose levels remain within normal limits. This compensatory mechanism is highly effective and can persist for years.
The problem is that this apparent stability masks progressive metabolic dysfunction. Elevated insulin promotes fat storage, suppresses fat oxidation, and alters appetite regulation. Over time, this creates a metabolic environment that favours weight gain, inflammation, and further insulin resistance. By the time fasting glucose finally begins to rise, the system has already endured prolonged stress, and the most reversible window has often passed.
Fasting insulin measures how much insulin the pancreas must produce to maintain glucose balance during a fasting state. Unlike glucose, insulin levels rise early in the course of metabolic dysfunction, long before any diagnostic thresholds are crossed. This makes fasting insulin a marker of metabolic effort rather than metabolic failure.
In a metabolically healthy individual, fasting insulin remains low because cells respond efficiently to insulin’s signal. After meals, insulin rises transiently and then returns to baseline as glucose is cleared. In insulin resistance, this baseline shifts upward. Insulin no longer returns to truly low levels, indicating that the system is under constant demand even in the absence of food intake.
Elevated fasting insulin tells a deeper story. It signals that cells are becoming resistant, that the pancreas is under chronic compensatory stress, and that metabolic flexibility, that is the ability to switch between fuel sources, is declining. Importantly, this state can exist for years without any abnormal glucose readings, making it invisible to standard screening.
At iThrive, fasting insulin is never interpreted in isolation. It is contextualised alongside clinical history, body composition, lipid patterns, inflammatory markers, and lifestyle factors. The goal is not merely to label insulin resistance, but to understand why insulin demand is rising in the first place.
Type 2 diabetes is often approached as a threshold-based diagnosis: glucose crosses a line, and disease is declared. In reality, diabetes unfolds along a continuum. Viewing insulin and glucose along a timeline reveals why their predictive value differs so dramatically.
In the earliest phase, insulin resistance begins to develop at the cellular level. Fasting insulin rises as the pancreas compensates, while fasting glucose remains normal. As dysfunction progresses, insulin levels climb higher and glucose may enter a borderline range. Eventually, pancreatic beta cells can no longer sustain compensation. Insulin output becomes inadequate relative to resistance, and glucose rises into the diabetic range.
This sequence explains why glucose is a late marker and insulin an early one. Relying on fasting glucose alone means intervening only after years of hidden metabolic stress have already occurred.
Both elevated fasting insulin and eventual glucose dysregulation arise from a shared underlying process: insulin resistance. Importantly, insulin resistance is not limited to blood sugar control. It is a systemic condition that affects multiple metabolic pathways simultaneously.
As insulin signalling weakens, lipid metabolism becomes dysregulated, promoting excess fat storage and impaired fat release. Inflammatory pathways are activated, mitochondrial efficiency declines, and appetite and satiety signalling become distorted. These changes explain why insulin resistance is often accompanied by weight gain, fatty liver, dyslipidaemia, PCOS, and chronic fatigue long before diabetes is diagnosed.
Despite growing evidence, fasting insulin is not routinely included in standard screening. This omission is not due to lack of relevance, but rather historical reliance on glucose-centric guidelines and a healthcare model oriented toward diagnosis rather than prevention.
For individuals with a family history of diabetes, persistent weight challenges, PCOS, or subtle metabolic symptoms, ignoring fasting insulin means missing the most actionable phase of disease development. By the time glucose rises, opportunities for early reversal are significantly reduced.
At iThrive, this missed window is precisely where root-cause assessment begins. Instead of asking whether sugar is high, we ask what the metabolism is signalling right now and why.
An elevated fasting insulin does not mean diabetes is inevitable. It means the system is under strain. The source of that strain determines both risk and reversibility.
Chronic overnutrition, sedentary muscle tissue, sleep disruption, stress-related cortisol elevation, inflammatory dietary patterns, and micronutrient deficiencies can all drive insulin resistance. Without identifying these drivers, focusing on numbers alone becomes misleading.
This is why Alive programmes prioritise mapping cause, compensation, and consequence. As discussed in our earlier piece on metabolic health restoration, numbers gain meaning only when interpreted within a broader physiological context.
Lowering fasting insulin can reduce diabetes risk, but only if the approach addresses the underlying drivers of insulin resistance. Superficial strategies that temporarily suppress glucose without improving insulin sensitivity may delay diagnosis but do not change disease trajectory.
Sustainable improvement requires enhancing muscle insulin sensitivity, reducing inappropriate hepatic glucose output, restoring metabolic flexibility, and correcting circadian and stress-related disruptions. This is where structured root-cause analysis becomes essential. For individuals uncertain about the origin of their insulin resistance, a clinical review can help clarify whether nutrition, stress physiology, inflammation, or hormonal patterns are the primary drivers.
Fasting insulin testing is particularly valuable for individuals whose glucose appears normal but whose metabolic health tells a different story. This includes those with a family history of type 2 diabetes, PCOS, fatty liver, unexplained weight gain, or early signs of insulin resistance.
For these individuals, waiting for glucose to rise is not prevention, rather it is postponement. Identifying metabolic strain early creates an opportunity to intervene before irreversible damage occurs.

Fasting glucose tells us when metabolic compensation has failed, but fasting insulin reveals when that compensation has begun. If the goal is to predict, prevent, and meaningfully reverse type 2 diabetes, insulin and not glucose offers the earlier and more actionable signal. True metabolic care requires moving beyond sugar thresholds toward understanding why insulin demand is rising in the first place. At iThrive, this shift from numbers to mechanisms allows intervention during the most reversible phase of metabolic dysfunction, long before irreversible damage sets in.
.webp)



.svg)
Diabetes is commonly explained as a problem of excess sugar in the blood. This explanation is simple, intuitive, and incomplete. Blood sugar elevation is not the starting point of diabetes, rather it is the final visible outcome of a much deeper collapse within the body’s metabolic infrastructure.
Long before glucose rises, the systems responsible for energy production, nutrient sensing, hormonal signaling, and cellular repair begin to malfunction. These systems form the body’s metabolic infrastructure. When this infrastructure weakens, the body loses its ability to process fuel efficiently, respond to insulin appropriately, and adapt to metabolic stress. Diabetes emerges only after years of silent dysfunction.
Understanding the root cause of diabetes requires moving beyond surface-level sugar metrics and examining how metabolic health deteriorates across tissues, organ systems, and time. This blog will explain why diabetes happens, what truly causes type 2 diabetes, and why addressing metabolic infrastructure failure is essential for meaningful prevention and reversal.
The root cause of diabetes is not sugar intake alone. If sugar were the primary cause, diabetes would appear rapidly and uniformly. Instead, it develops slowly, selectively, and progressively. This pattern indicates a failure of internal regulation rather than simple exposure.
Diabetes root cause lies in the body’s declining ability to manage energy. Metabolic infrastructure includes mitochondrial function, insulin signaling pathways, hormonal coordination, nutrient sensing, circadian alignment, and cellular repair mechanisms. These systems collectively determine how efficiently the body processes glucose, fats, and proteins.
When metabolic infrastructure is intact, the body can tolerate dietary variation and metabolic stress. When it fails, even modest demands overwhelm regulatory capacity, leading to chronic hyperinsulinemia, insulin resistance, and eventually elevated blood sugar.
To understand what causes diabetes, we must examine how multiple systems fail together. Diabetes is not caused by a single organ malfunctioning in isolation. It is the outcome of progressive system-wide inefficiency.
Mitochondria lose efficiency due to chronic overnutrition, inflammation, and nutrient deficiencies. Insulin signaling pathways become desensitized from constant stimulation. Hormonal rhythms lose synchrony due to circadian disruption and stress. Cellular repair mechanisms fall behind ongoing damage.
Each failure compounds the next. The body compensates by increasing insulin output, diverting energy into fat storage, and suppressing fat oxidation. Over time, compensation becomes unsustainable.
Metabolic infrastructure is not a metaphor. It refers to concrete biological systems that govern energy flow. This includes mitochondria that generate ATP, enzymes that process nutrients, receptors that transmit hormonal signals, and cellular membranes that regulate transport.
Insulin resistance arises when these systems lose efficiency. Cells become energetically overloaded yet functionally starved. Glucose remains in circulation not because it is abundant, but because cells cannot utilize it properly.
Metabolic dysfunction therefore reflects an inability to convert nutrients into usable energy. This distinction explains why reducing sugar intake alone rarely restores metabolic health once infrastructure has degraded.

High blood sugar appears only after years of metabolic decline. During early dysfunction, insulin levels rise to maintain glucose control. As insulin resistance worsens, glucose tolerance narrows. Eventually, glucose spills into circulation.
By the time diabetes is diagnosed, metabolic infrastructure has already been compromised across multiple systems. Treating sugar at this stage is reactive, not preventive.
This is why diabetes causes explained solely through glucose fail to capture disease biology. Sugar is a symptom, not the origin.
Causes of type 2 diabetes extend far beyond food choices. Chronic psychological stress elevates cortisol, increasing glucose production and impairing insulin signaling. Sleep deprivation disrupts hormonal regulation and mitochondrial repair.
Circadian misalignment alters nutrient partitioning, increasing insulin resistance even with normal caloric intake. Environmental toxins interfere with endocrine signaling. Micronutrient deficiencies impair enzymatic reactions essential for glucose metabolism.
These inputs degrade metabolic infrastructure gradually, explaining why individuals with similar diets experience vastly different outcomes.

At its core, diabetes reflects energy mismanagement. Cells are exposed to abundant fuel but lack the capacity to process it efficiently. Excess energy is diverted into fat storage, inflammation, and oxidative stress.
Insulin becomes a compensatory signal rather than a regulatory one. Metabolic health declines as flexibility is lost. The body becomes locked into inefficient pathways that prioritize short-term survival over long-term stability.
This perspective aligns with Alive’s focus on early metabolic screening and infrastructure repair rather than glucose suppression alone.
Restoring metabolic health requires repairing infrastructure, not just controlling outputs. This involves improving mitochondrial efficiency, reducing chronic insulin demand, restoring circadian rhythm, and correcting nutrient deficiencies.
Strategic fasting allows metabolic systems to reset and clear accumulated metabolic stress. Sleep and stress regulation restore hormonal coordination. Physical activity improves glucose uptake independent of insulin.
This root-cause approach differs fundamentally from symptom management. It focuses on why diabetes happens rather than how to temporarily control it.
For individuals unsure where dysfunction lies, a Root Cause Analysis helps identify which components of metabolic infrastructure are compromised. You may also choose to book a free consult to determine whether deeper evaluation is appropriate.

Waiting for diabetes diagnosis means waiting for infrastructure collapse. Early screening that includes insulin dynamics, metabolic markers, and lifestyle assessment identifies dysfunction when reversal is still possible.
This is why Alive prioritizes early detection over late intervention. When metabolic health is restored early, glucose often normalizes as a downstream effect.
The root cause of diabetes is not elevated blood sugar but the gradual failure of the body’s metabolic infrastructure. Long before glucose rises, mitochondrial inefficiency, insulin resistance, hormonal disruption, and circadian misalignment erode metabolic health. Treating sugar addresses a late-stage symptom, while repairing infrastructure targets the disease itself. Sustainable diabetes prevention and reversal depend on restoring the systems that regulate energy, not merely controlling its byproducts.




.svg)
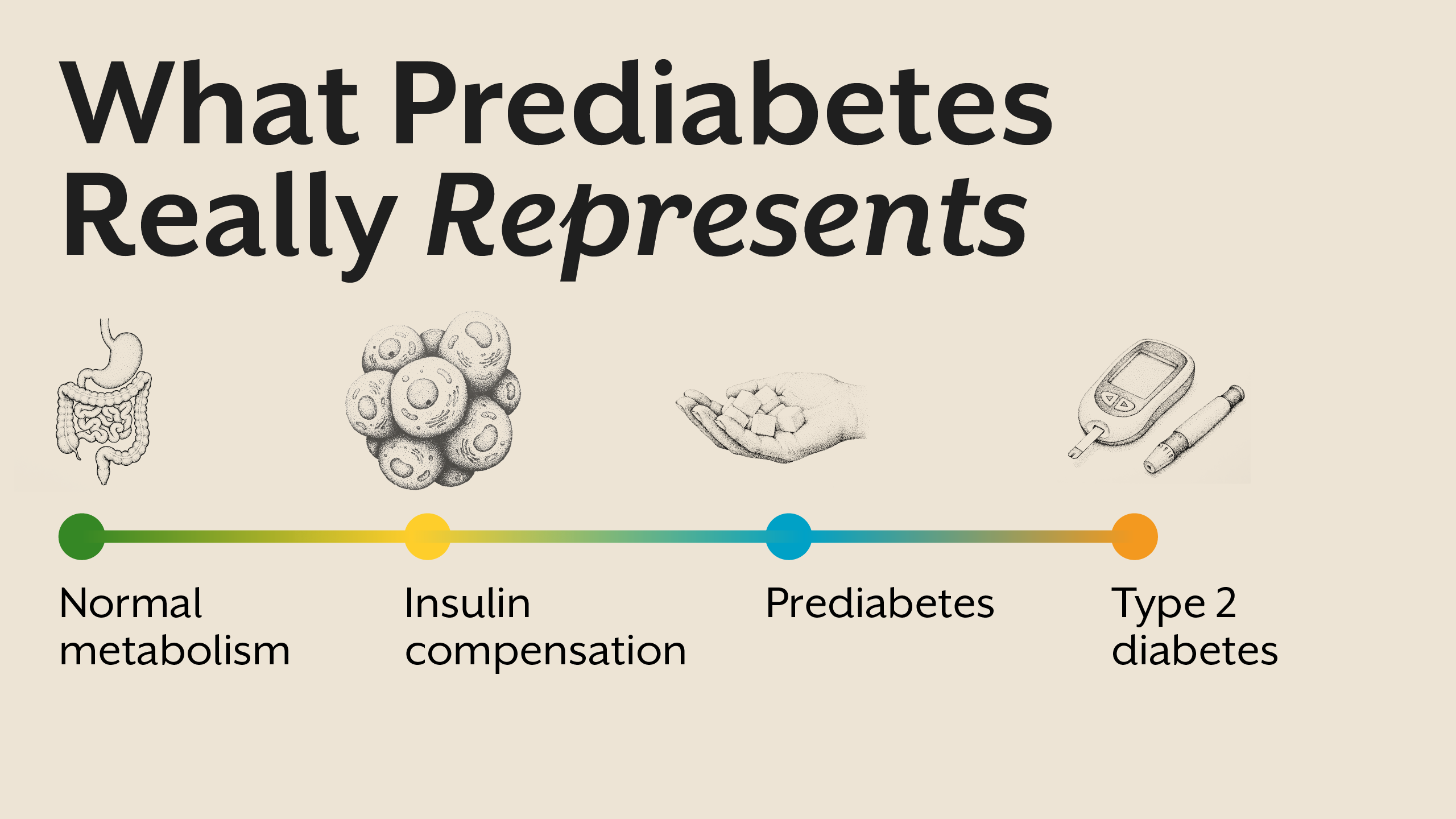
Prediabetes is often presented as a mild elevation in blood sugar, a temporary phase between “normal” and diabetes. This framing is misleading and, in many cases, dangerously incomplete. Prediabetes is not a glucose problem waiting to become severe. It is a metabolic warning signal indicating that the body’s energy regulation systems are already under strain.
Most individuals diagnosed with prediabetes are told to “watch sugar,” “eat less carbs,” or “exercise more.” While these suggestions are not incorrect, they fail to address the underlying physiology driving the condition. Blood sugar rises only when the body can no longer compensate for deeper metabolic dysfunction, particularly insulin resistance.
Understanding what prediabetes truly represents requires shifting focus away from sugar alone and toward metabolic health as a whole. This blog will explain the real meaning of prediabetes, why it develops, how insulin resistance sits at its core, and why early metabolic screening matters far more than waiting for diabetes to appear.
Prediabetes meaning is often simplified to “slightly high blood sugar.” Clinically, it is defined by fasting glucose, post-meal glucose, or HbA1c values that fall above normal but below diabetic thresholds. However, these numbers reflect outcomes, not causes.
Prediabetes represents a state where the body is struggling to maintain glucose balance despite increasing hormonal effort. Insulin levels are often elevated, signaling that cells are becoming less responsive. Blood sugar rises not because sugar intake suddenly increases, but because insulin’s effectiveness is declining.
In this phase, glucose may still appear controlled at rest, but metabolic flexibility is impaired. The body has lost its ability to adapt efficiently to meals, fasting, stress, and physical activity. Prediabetes is therefore not a borderline condition; it is an early disease state.
Sugar does not create prediabetes in isolation. If it did, every individual consuming carbohydrates would develop the condition. The real issue lies in how the body processes and responds to glucose.
Insulin resistance is the central driver. When cells fail to respond efficiently to insulin, glucose remains in circulation longer, prompting the pancreas to release more insulin. Over time, this compensatory mechanism becomes insufficient.
Additional contributors include chronic stress, sleep deprivation, circadian disruption, inflammation, micronutrient deficiencies, and frequent eating without metabolic rest. These factors collectively impair insulin signaling and mitochondrial efficiency.
Prediabetes symptoms are not dramatic. Most individuals do not feel “unwell” enough to seek medical attention. Instead, they experience gradual changes that are normalized or ignored.
Common symptoms include persistent fatigue, post-meal drowsiness, difficulty losing weight, increased hunger, sugar cravings, and abdominal fat gain. Cognitive symptoms such as brain fog and reduced focus are also common.
Because blood sugar levels are only mildly elevated, these symptoms are rarely linked to metabolic dysfunction. Yet they reflect a body working harder to maintain balance.
Insulin resistance sits at the heart of prediabetes. It develops when cells in muscle, liver, and adipose tissue stop responding effectively to insulin’s signal. As resistance increases, insulin levels rise to compensate.
This process is not uniform. Muscle tissue may resist insulin first, impairing glucose uptake. The liver may continue producing glucose despite insulin presence. Adipose tissue may remain insulin sensitive, promoting fat storage.
These tissue-specific responses explain why individuals with prediabetes often gain visceral fat while still being told their sugar is “borderline.” Insulin resistance reflects systemic metabolic dysfunction, not a single lab value.

Prediabetes causes are cumulative, not isolated. Frequent eating keeps insulin elevated throughout the day. Chronic stress elevates cortisol, worsening glucose production and insulin resistance. Poor sleep impairs insulin signaling and mitochondrial function.
Circadian disruption from late nights and irregular meals further destabilizes metabolic rhythms. Nutrient deficiencies, particularly magnesium and B-complex vitamins, impair glucose metabolism at the cellular level.
Prediabetes reflects a system under constant demand without sufficient recovery. Addressing it requires restoring metabolic rhythm, not simply lowering sugar intake.
Prediabetes treatment is often framed as glucose reduction. However, targeting sugar alone ignores the hormonal dysfunction driving the condition.
Effective intervention focuses on improving insulin sensitivity, reducing insulin demand, restoring circadian alignment, and supporting mitochondrial health. Meal timing, sleep quality, stress regulation, and nutrient sufficiency play central roles.
Strategic fasting, when individualized, allows insulin levels to normalize and receptors to resensitize. Physical activity improves glucose uptake independent of insulin. These approaches address root causes rather than symptoms.

Prediabetes is often treated as a warning label with no urgency. In reality, it is a narrow window where metabolic flexibility can still be restored.
Once insulin resistance progresses and pancreatic capacity declines, reversal becomes significantly harder. Early identification allows targeted intervention before irreversible damage occurs.
This is why Alive emphasizes early metabolic screening and Root Cause Analysis rather than waiting for diagnostic thresholds. If you are uncertain whether prediabetes reflects deeper metabolic dysfunction, you may consider booking a free consult to understand whether further evaluation is needed.
Prediabetes is not an isolated diagnosis. It reflects declining metabolic health across systems. Improving metabolic health improves glucose regulation as a downstream effect.
This broader view aligns with Essentials philosophy, where foundational metabolic support precedes symptom management. When insulin sensitivity improves, glucose stability often follows naturally.
Prediabetes is not a mild sugar abnormality but an early warning of metabolic dysfunction driven primarily by insulin resistance. Blood glucose rises only after the body has exhausted years of hormonal compensation, making prediabetes a critical window for intervention rather than a passive waiting phase. Addressing metabolic health through insulin sensitivity, lifestyle rhythm, and root cause evaluation allows meaningful reversal before long-term disease develops.



.svg)
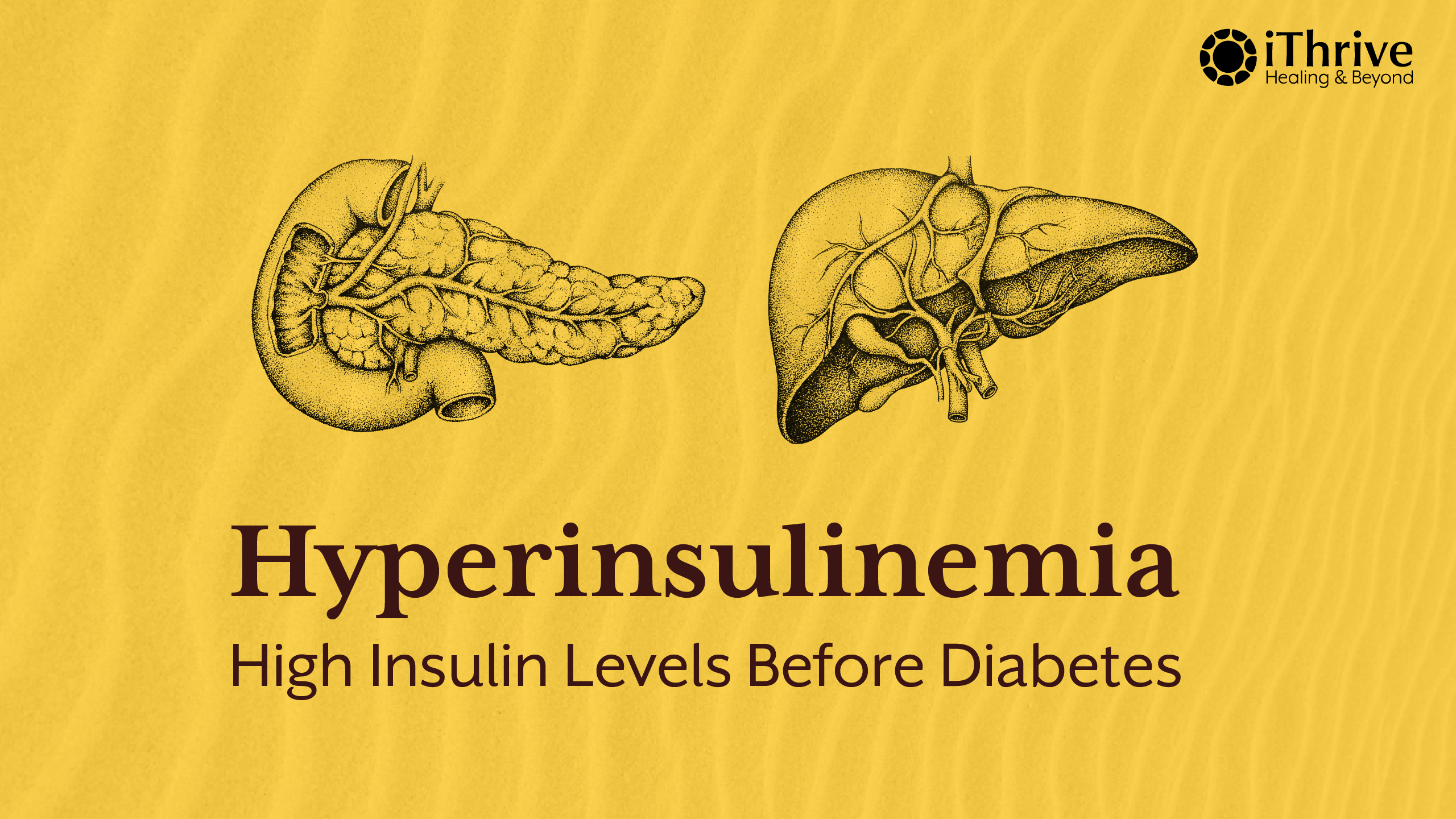
Modern metabolic disease rarely begins with high blood sugar. Long before glucose levels cross diagnostic thresholds, the body is already compensating, adapting, and quietly struggling to maintain balance. At the center of this compensation lies insulin.
Hyperinsulinemia refers to a state of chronically elevated insulin levels in the bloodstream. It is one of the earliest detectable signs of metabolic dysfunction, yet it remains largely invisible in routine healthcare. Most individuals with hyperinsulinemia are told their blood sugar is “normal” and reassured that nothing is wrong, even as their metabolism is under increasing strain.
This silent elevation of insulin is not benign. It drives insulin resistance, alters fat storage, disrupts hormonal signaling, and lays the foundation for conditions such as type 2 diabetes, fatty liver disease, PCOS, and cardiovascular disease. Understanding hyperinsulinemia requires shifting attention away from glucose alone and toward the hormonal systems regulating energy.
This blog will explore what hyperinsulinemia is, how it develops, why it goes undetected, how it manifests in the body, and why identifying it early changes the entire trajectory of metabolic health.
Hyperinsulinemia is defined by persistently high insulin levels relative to physiological need. Insulin is secreted by the pancreas in response to rising blood glucose, but its role extends far beyond glucose control. It signals cells to store energy, suppress fat breakdown, and shift metabolism toward growth and storage.
In a healthy system, insulin rises briefly after meals and returns to baseline during fasting. In hyperinsulinemia, insulin remains elevated for prolonged periods, including during fasting. This elevation reflects a loss of metabolic efficiency, where the body must produce more insulin to achieve the same effect.
Importantly, hyperinsulinemia can exist even when fasting glucose and HbA1c remain within reference ranges. Blood sugar appears controlled only because insulin is working overtime. This distinction is critical, as glucose tests alone do not reveal the hormonal cost of maintaining that control.
Over time, chronically high insulin alters receptor sensitivity, cellular signaling, and metabolic flexibility, setting the stage for insulin resistance.

Insulin is often viewed as protective because it lowers blood glucose. However, chronically elevated insulin represents a state of metabolic stress. Cells exposed to high insulin for extended periods reduce receptor responsiveness as a protective mechanism, leading to insulin resistance.
High insulin levels also shift the body into a persistent storage mode. Fat breakdown is suppressed, lipogenesis increases, and metabolic flexibility declines. This makes weight loss increasingly difficult, even with calorie restriction.
Beyond metabolism, hyperinsulinemia influences inflammation, oxidative stress, and vascular function. Elevated insulin stimulates sympathetic nervous system activity, contributing to hypertension, and alters lipid metabolism, promoting atherogenic profiles.
Rather than preventing disease, prolonged hyperinsulinemia actively drives it.
Despite insulin’s central role in metabolic health, insulin levels are not routinely tested. Standard screening focuses on fasting glucose, post-prandial glucose, and HbA1c. These markers assess glycemia, not insulin dynamics.
The absence of insulin testing stems from historical disease models that defined diabetes by glucose thresholds rather than hormonal dysfunction. As long as glucose remains normal, insulin compensation goes unnoticed.
The insulin levels test, particularly the fasting insulin test, offers insight into how hard the pancreas is working to maintain glucose balance. Without this data, early metabolic dysfunction is often missed.
This gap explains why many individuals are diagnosed with insulin resistance or type 2 diabetes only after years of silent progression.
Hyperinsulinemia and insulin resistance reinforce each other in a self-perpetuating cycle. As cells become less responsive to insulin, the pancreas compensates by secreting more. This increased insulin exposure further reduces sensitivity, accelerating resistance.
This process does not occur uniformly. Liver cells, muscle tissue, and adipose tissue develop insulin resistance at different rates. The liver may continue producing glucose despite high insulin, while adipose tissue remains sensitive, promoting fat storage.
This tissue-specific resistance explains why individuals can experience fat gain, fatigue, and metabolic dysfunction long before glucose levels rise. Insulin resistance is therefore not a binary state but a spectrum of dysfunction across systems.
High insulin symptoms are often nonspecific, which contributes to underrecognition. Individuals may experience chronic fatigue, increased hunger shortly after meals, difficulty losing weight, brain fog, and afternoon energy crashes.
Other insulin resistance symptoms include increased abdominal fat, sugar cravings, poor exercise recovery, and disrupted sleep. In women, hyperinsulinemia may worsen androgen excess and menstrual irregularities. In men, it may contribute to reduced metabolic drive and visceral fat accumulation.
Because these symptoms overlap with stress, aging, or lifestyle issues, they are rarely linked back to insulin dysregulation in clinical settings.

Insulin resistance does not arise from a single dietary choice. It reflects cumulative metabolic strain from multiple inputs. Frequent eating without metabolic rest keeps insulin elevated throughout the day, preventing baseline recovery.
Chronic psychological stress increases cortisol, which raises glucose output and worsens insulin resistance. Sleep deprivation impairs insulin signaling and mitochondrial function. Circadian misalignment disrupts hormonal rhythms essential for glucose handling.
Nutrient deficiencies, particularly magnesium, chromium, and B vitamins, impair insulin signaling pathways. Gut-derived inflammation further interferes with receptor function.
Understanding insulin resistance causes requires a systems-level lens rather than isolated blame on sugar or calories.
The fasting insulin test is one of the most valuable yet underutilized tools in metabolic assessment. It reflects baseline insulin demand and provides early insight into compensation.
Normal insulin levels vary across labs, but optimal fasting insulin is generally lower than what is considered “acceptable.” Elevated fasting insulin indicates that insulin resistance is already present, even if glucose remains normal.
Pairing fasting insulin with fasting glucose allows assessment of insulin sensitivity and metabolic efficiency. Tracking trends over time is often more informative than single measurements.
IThrive’s screening model prioritizes these early signals to identify dysfunction before irreversible damage occurs. This approach is discussed further in our Academy content on fasting insulin and early metabolic screening.

Insulin resistance treatment is most effective when it reduces insulin demand rather than forcing glucose down. This requires restoring insulin sensitivity, not overriding physiology.
Dietary strategies focus on reducing glycemic load, improving meal timing, and allowing sufficient fasting periods. Strategic fasting, when individualized, lowers baseline insulin and allows receptor resensitization.
Lifestyle interventions such as sleep optimization, stress regulation, and circadian alignment directly improve insulin signaling. Nutrient repletion supports mitochondrial function and glucose oxidation.
This root-cause approach contrasts with glucose-centric management, which often leaves hyperinsulinemia unaddressed.
Hyperinsulinemia is not a neutral state. Chronic high insulin accelerates fat accumulation, promotes inflammation, and increases cardiometabolic risk independent of glucose levels.
It is strongly associated with obesity, fatty liver disease, hypertension, dyslipidemia, and cognitive decline. In many cases, type 2 diabetes represents the late manifestation of years of insulin overload.
Identifying and addressing hyperinsulinemia early preserves metabolic flexibility and reduces long-term disease risk.
For those seeking clarity on whether insulin resistance or hyperinsulinemia may already be present, a root cause analysis can provide direction. You may choose to book a free consult to understand whether deeper metabolic evaluation is appropriate.
Hyperinsulinemia represents one of the earliest and most overlooked signals of metabolic dysfunction, often developing years before blood sugar levels become abnormal. Chronically high insulin levels reflect a state of compensation, not metabolic health, and quietly drive insulin resistance, fat accumulation, inflammation, and long-term cardiometabolic risk. Because routine screening rarely includes insulin testing, many individuals remain unaware of this condition until later stages of disease emerge. Measuring fasting insulin and addressing the underlying drivers of insulin resistance allows intervention at a stage where metabolic flexibility can still be restored, shifting care from late-stage management to true root-cause resolution.


A complete blood panel of 60+ parameters, collected from your home by a phlebotomist. Analysis by a dedicated functional nutritionist using optimal ranges, not the standard reference ranges your doctor uses. Your symptoms, lifestyle, and health history factored into the analysis. And a 60 to 90-minute one-on-one video consultation where your nutritionist walks you through everything they found. The blood tests are included in the price. There are no hidden charges or extra cost for anything.

A complete blood panel of 60+ parameters, collected from your home by a phlebotomist. Analysis by a dedicated functional nutritionist using optimal ranges, not the standard reference ranges your doctor uses. Your symptoms, lifestyle, and health history factored into the analysis. And a 60 to 90-minute one-on-one video consultation where your nutritionist walks you through everything they found. The blood tests are included in the price. There are no hidden charges or extra cost for anything.

A complete blood panel of 60+ parameters, collected from your home by a phlebotomist. Analysis by a dedicated functional nutritionist using optimal ranges, not the standard reference ranges your doctor uses. Your symptoms, lifestyle, and health history factored into the analysis. And a 60 to 90-minute one-on-one video consultation where your nutritionist walks you through everything they found. The blood tests are included in the price. There are no hidden charges or extra cost for anything.

A complete blood panel of 60+ parameters, collected from your home by a phlebotomist. Analysis by a dedicated functional nutritionist using optimal ranges, not the standard reference ranges your doctor uses. Your symptoms, lifestyle, and health history factored into the analysis. And a 60 to 90-minute one-on-one video consultation where your nutritionist walks you through everything they found. The blood tests are included in the price. There are no hidden charges or extra cost for anything.

A complete blood panel of 60+ parameters, collected from your home by a phlebotomist. Analysis by a dedicated functional nutritionist using optimal ranges, not the standard reference ranges your doctor uses. Your symptoms, lifestyle, and health history factored into the analysis. And a 60 to 90-minute one-on-one video consultation where your nutritionist walks you through everything they found. The blood tests are included in the price. There are no hidden charges or extra cost for anything.

A complete blood panel of 60+ parameters, collected from your home by a phlebotomist. Analysis by a dedicated functional nutritionist using optimal ranges, not the standard reference ranges your doctor uses. Your symptoms, lifestyle, and health history factored into the analysis. And a 60 to 90-minute one-on-one video consultation where your nutritionist walks you through everything they found. The blood tests are included in the price. There are no hidden charges or extra cost for anything.

A complete blood panel of 60+ parameters, collected from your home by a phlebotomist. Analysis by a dedicated functional nutritionist using optimal ranges, not the standard reference ranges your doctor uses. Your symptoms, lifestyle, and health history factored into the analysis. And a 60 to 90-minute one-on-one video consultation where your nutritionist walks you through everything they found. The blood tests are included in the price. There are no hidden charges or extra cost for anything.



